When plastic enters the cell
The intracellular fate of microplastics and nanoplastics after endocytosis
One of the questions we are asked most often is simple.
If microplastics enter our bodies, what happens to them once they are inside our cells?
It is a reasonable question. Cells constantly break things down. They recycle damaged proteins, digest invading microbes, and dismantle old cellular components. Because of that, many people assume that if a plastic particle enters a cell, the cell will eventually digest it.

The scientific literature suggests the story is more complicated.
A growing body of peer reviewed research shows that when microplastics and nanoplastics are taken up by cells, they do not simply disappear into the background of the cell. In macrophages and barrier epithelial cells, the most common fate is endocytic or phagocytic uptake followed by trafficking into the endo-lysosomal system, a network of vesicles responsible for sorting and processing internalized material [1-3].
Across many experimental systems, the most consistent observation is not rapid destruction of the plastic polymer itself. Instead, researchers frequently observe particle persistence accompanied by signs of cellular stress, particularly involving lysosomes, autophagy pathways, mitochondria, and inflammatory signaling networks [2,4-6].
The literature also makes an important distinction between where particles go inside a cell and whether they are actually broken down there.
For common commodity plastics dominated by carbon-carbon backbones, including polyethylene (PE), polypropylene (PP), polystyrene (PS), and polyvinyl chloride (PVC), current evidence suggests that once inside cells they tend to persist with only limited chemical transformation. By contrast, polymers that contain hydrolyzable ester bonds, such as PLA, PLGA, PCL, and some polyurethane systems, are more chemically compatible with acidic intracellular compartments and esterase activity. These materials are therefore more likely to undergo partial breakdown in cells [7-10].
Taken together, the emerging scientific picture is measured but consistent.
Many plastic particles can enter cells, and many are ultimately routed into lysosomes. For most common environmental plastics, efficient intracellular breakdown appears limited. As particles accumulate inside cells, their presence may begin to influence normal cellular behavior[4,11-13].
Why this question matters
A common assumption is that if a plastic particle enters a cell, the cell will eventually digest it.
Current evidence does not support that as a general rule.
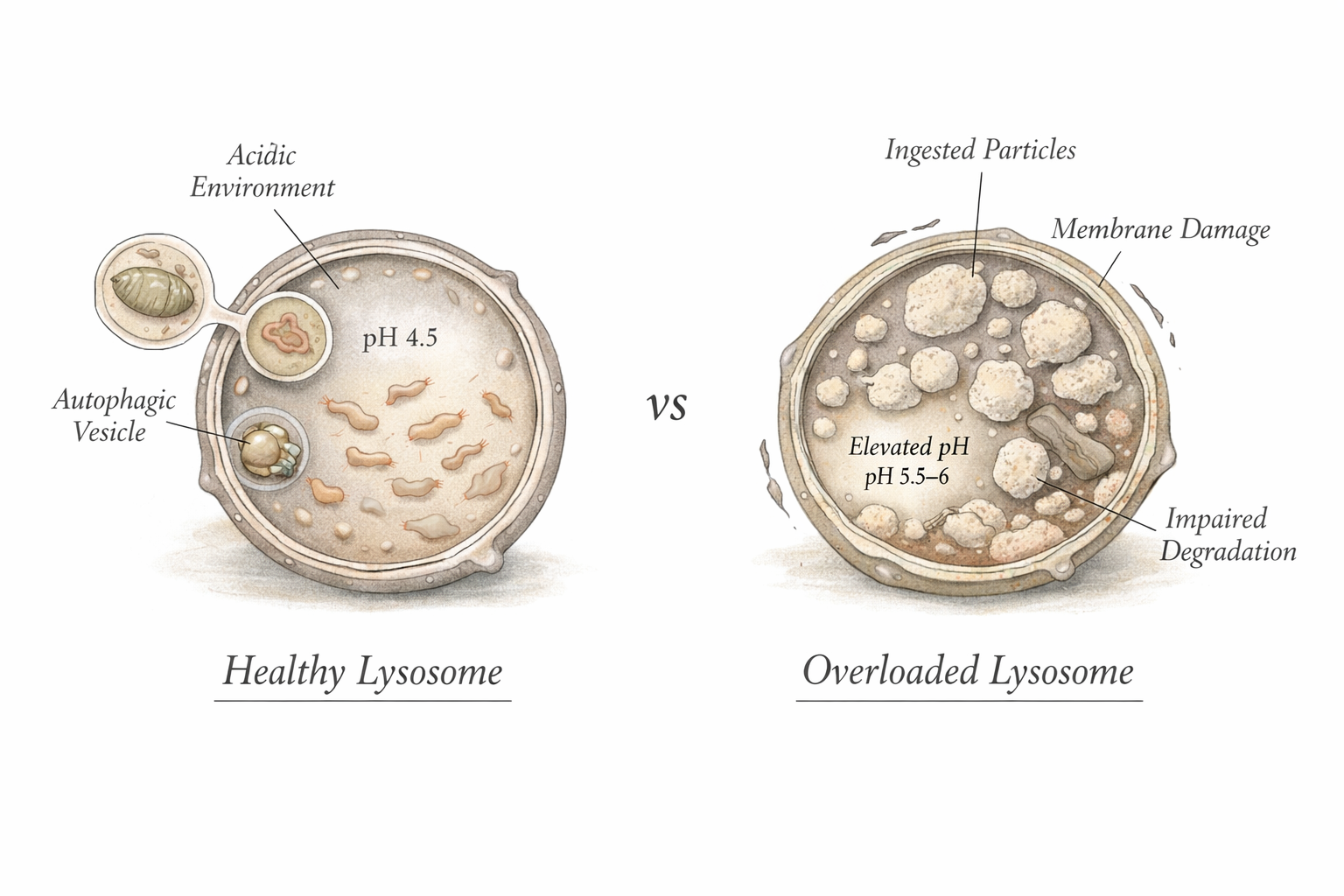
Inside most cells is a compartment called the lysosome. Lysosomes are sometimes described as the recycling centers of the cell. They maintain an acidic internal environment and contain dozens of enzymes capable of breaking down biological molecules such as proteins, lipids, and nucleic acids [7,14].
However, lysosomes evolved to process biological chemistry, not modern synthetic materials.

Most plastics are built from extremely stable chemical bonds that do not resemble the bonds found in natural biological molecules. As a result, many plastic polymers are not recognized as easy substrates for lysosomal enzymes.
That mismatch matters.
It means that when a plastic particle enters a cell, the cell’s recycling machinery may not be able to fully dismantle it. Instead, the particle may remain inside intracellular compartments for extended periods.
This distinction is important. Exposure is one question. Cellular fate is another. The fact that a particle has been endocytosed does not necessarily mean it has been neutralized.
In several cellular studies, biological responses appear to scale more closely with the amount of plastic that accumulates inside the cell than with the nominal plastic concentration in the surrounding medium [2,15].
In other words, what may matter biologically is not only how much plastic exists outside a cell, but how much ultimately builds up inside it.
The central finding
Across many experimental systems, the most consistent intracellular destination for microplastics and nanoplastics is the endo-lysosomal system.
After endocytosis in epithelial cells or phagocytosis in macrophages, particles are commonly observed in early endosomes, late endosomes, phagosomes, phagolysosomes, and lysosomes. Lysosomal colocalization has been supported through fluorescence imaging, ultrastructural microscopy, and functional lysosomal assays [1,16-18].
This does not mean that every particle enters cells in the same way.

Particle size, surface chemistry, and shape can influence the pathway used for uptake:
- smaller nanoparticles often enter through clathrin mediated endocytosis
- intermediate sized particles may engage caveolar or lipid raft pathways
- irregular particles can enter through macropinocytosis
- micron-scale plastics are especially relevant to phagocytosis in macrophages [16-20].
Confocal microscopy studies help illustrate how these uptake processes appear inside real cells. Researchers exposed macrophages to fluorescent polystyrene particles [2]. Control cells showed no internal signal (panel A, left), while exposed cells displayed clear particle accumulation within the cytoplasm. Smaller nanoplastic beads (40–90 nm) appeared as dispersed intracellular puncta (panel B, middle), whereas larger particles (0.7–0.9 μm) were visible as discrete internalized bodies consistent with phagocytic uptake (panel C, right). These images confirm that plastic particles are not merely attached to the cell surface but can be actively internalized by macrophages across a range of particle sizes.
Despite these differences in entry routes, many of these pathways eventually converge on lysosomes unless particles escape endosomes or are exported through exocytic processes [18,21].
What happens after uptake
Once plastic particles are inside the cell, several patterns appear repeatedly across the scientific literature.
Particle persistence
One of the most common observations is that plastic particles remain inside cells for extended periods of time.
Multiple macrophage studies report that internalized polystyrene microbeads or nanoplastics are not efficiently digested and can remain inside cells while influencing cellular behavior [2,15,21].
Several studies have shown that once plastic particles are internalized, they can remain inside cells for extended periods. In one macrophage experiment, cells exposed to fluorescent polystyrene beads were analyzed immediately after a 24 hour exposure and again after a nine day recovery period [2]. Confocal microscopy confirmed that the particles were internalized rather than simply attached to the cell surface. Uptake was strongly size dependent. Smaller particles were internalized by nearly all macrophages, with roughly 90 to 99 percent of cells containing particles in the 40 to 90 nm and 0.7 to 0.9 μm ranges. As particle size increased, uptake declined sharply. Around 2 μm, only about half of macrophages contained particles, and for very large particles in the 6 to 8 μm range only about 9 percent of cells showed internalization.
Follow up measurements after the nine day recovery period showed that intracellular fluorescence remained largely stable. Although mean fluorescence intensity per cell decreased slightly, the total signal across cells remained similar, consistent with dilution of particles as macrophages divided rather than degradation of the particles themselves. Together, these observations support the view that once internalized, plastic particles can persist inside macrophages rather than being rapidly dismantled by cellular digestion pathways.
Lysosomal stress
Because many particles accumulate in lysosomes, signs of lysosomal stress are frequently reported. These include impaired lysosomal acidification, altered activity of lysosomal enzymes such as cathepsins, lysosomal membrane permeabilization, vacuolization of lysosomal compartments, reduced numbers of functional acidic lysosomes, impaired autophagic flux [4,5,22-24].
These changes suggest that lysosomes can become overloaded or destabilized when plastic particles accumulate inside them.

Downstream organelle effects
Cells are highly interconnected systems. When lysosomes become stressed, effects can spread to other organelles.
Researchers frequently report downstream changes in mitochondria, including loss of mitochondrial membrane potential, increased mitochondrial reactive oxygen species, altered cellular respiration, activation of inflammatory signaling pathways [6,25-27]
Evidence for plastic particles consistently localizing beyond lysosomes is much less certain.
Some studies report signals suggesting particles inside the endoplasmic reticulum or nucleus. However, these observations can be difficult to interpret because fluorescent labels can leak from particles, lysosomal environments can distort fluorescence signals, and microscopy artifacts can produce misleading colocalization patterns [3,28-31].
For this reason, the most cautious interpretation today is that lysosomal localization is strongly supported, while direct localization inside the nucleus, mitochondrial interior, or peroxisomal lumen remains uncertain.
Can cells break plastics down
The answer depends strongly on polymer chemistry.
For plastics dominated by carbon-carbon backbones, such as polyethylene, polypropylene, polystyrene, and polyvinyl chloride, true intracellular depolymerization appears limited. These materials are chemically resistant, and mammalian cells do not possess a general enzymatic toolkit capable of efficiently cleaving them into small metabolites [11,12,32-35].
Intracellular chemistry may still modify particle surfaces. Possible transformations include, formation of carbonyl groups, partial disruption of aromatic structures, changes in particle brittleness, altered protein corona behavior, and release of adsorbed additives [32,36,37]
However, surface oxidation is not the same as efficient polymer destruction.
For plastics containing hydrolyzable ester bonds, the situation is different. Polymers such as PLA, PLGA, PCL, and certain polyurethane materials are chemically compatible with acidic lysosomes and intracellular esterase activity. In these systems, studies report partial degradation and release of smaller breakdown products over time, especially for small or amorphous particles [8-10,38].
This highlights an important principle.
The key factor is not whether a material is called a plastic, but whether its chemical bonds can be accessed by mammalian intracellular chemistry.
| Polymer | Chemistry | Likely intracellular fate |
| PE / PP | Mostly carbon-carbon backbone | Surface oxidation or minor chemical modification may occur, but efficient intracellular depolymerization appears unlikely in mammalian cells |
| PS | Carbon backbone with aromatic side groups | Intracellular persistence appears more likely than efficient degradation; surface chemistry and redox interactions may contribute to cellular stress responses |
| PET | Aromatic ester backbone | Can be depolymerized by specialized microbial enzymes, but mammalian intracellular conditions appear insufficient for efficient polymer breakdown |
| PLA / PLGA / PCL | Aliphatic ester rich polymers | More chemically compatible with acidic lysosomal environments and esterase activity, allowing partial intracellular degradation over time |
Lysosomes, mitochondria, and peroxisomes
Lysosomes
Lysosomes are the primary intracellular compartment involved in plastic particle processing. Their acidic environment is maintained by V-ATPase proton pumps and associated ion conductances, enabling the activity of numerous acid hydrolases [7,14].
For hydrolyzable polymers, lysosomes may act as a degradative compartment.
For most environmental plastics, however, lysosomes appear to function more as a holding compartment than a disposal system [4,22].
Mitochondria
Mitochondria are often affected even when direct particle localization inside mitochondria is not demonstrated. The more reproducible observation is functional spillover following lysosomal stress, including increased reactive oxygen species, altered membrane potential, and impaired respiration [6,25,39].
Peroxisomes
Peroxisomes are redox active organelles involved in hydrogen peroxide metabolism and lipid signaling. They are therefore biologically relevant to oxidative stress pathways. However, evidence for physical accumulation of plastic particles inside peroxisomes remains limited. Current research more strongly supports peroxisome related pathway disruption rather than direct particle localization within peroxisomes [13,40].
What happens when particle burden builds up
When intracellular particle burden increases, several biological responses appear repeatedly across studies.
One is lysosome centered dysfunction.
Another is autophagy disruption, where damaged or overloaded lysosomes impair normal cellular recycling processes and lead to accumulation of autophagosomes or p62 associated signaling signatures [23,24].
A third is immunometabolic reprogramming. In macrophages, plastic exposure has been associated with shifts toward glycolysis, reduced mitochondrial respiration, and changes in immune activation markers, suggesting that particle accumulation can influence how immune cells allocate energy and respond to environmental signals [20,41].
A fourth is inflammatory amplification. In polystyrene nanoparticle systems in particular, lysosomal injury and reactive oxygen species have been linked to NLRP3 inflammasome signaling, although the magnitude of this effect varies depending on particle chemistry, dose, and experimental model [6,26,27].

This does not mean every internalized particle causes meaningful biological harm in every context.
Particle size, surface chemistry, aging state, additives, and the specific cell type involved all influence outcomes [11,13,42].
However, the broader pattern across studies is notable: intracellular accumulation is not biologically silent.
What the field still does not know
Despite rapid progress, several important gaps remain.
One of the largest is chemical fate. Many studies show where particles localize and how cells respond, but relatively few recover intracellular particles and directly measure whether polymer molecular weight declines, oxidation products form, or monomers are released [11,32,43].
Another limitation involves material realism. Much mechanistic work still relies on pristine, monodisperse polystyrene spheres. Real world exposures involve mixed polymers, irregular particle shapes, weathering, biofilms, and additives. Studies comparing pristine and environmentally aged plastics suggest that particle aging can significantly alter lysosomal and autophagic outcomes [4,23].
Finally, methodological artifacts remain a challenge. Fluorescent labels can leach from particles. Lysosomal environments can distort fluorescence signals. Some staining methods can introduce false positives. As a result, newer studies increasingly rely on label free Raman approaches and orthogonal detection techniques to improve accuracy [2-3,29-31,44].
Conclusion
The most measured conclusion is also the most important.
After endocytosis or phagocytosis, microplastics and nanoplastics are commonly routed into the endo-lysosomal system. In many cases they persist there, sometimes with slow exocytic clearance, but often without convincing evidence of efficient intracellular depolymerization for the major commodity plastics that dominate modern exposure [1,2,11,18].
What follows can be biologically active.
Across studies, intracellular plastic burden has been associated with lysosomal dysfunction, autophagy disruption, mitochondrial stress, oxidative signaling, and inflammatory responses, particularly as internal particle load increases [4-6,22-24,25].
The field still needs better chemistry measurements, more realistic particle models, and improved organelle-resolved tracking.
But the evidence already supports a careful conclusion.
Once plastics enter cells, they may remain there longer than the cell can comfortably ignore. For many common polymers, intracellular persistence currently appears more plausible than efficient intracellular destruction.
References
- 1.↑ Brandts, I. et al. Polystyrene nanoplastics target lysosomes interfering with lipid metabolism through the PPAR system and affecting macrophage functionalization. Environ. Sci.: Nano 10, 2245-2258 (2023).
- 2.↑ Collin-Faure, V. et al. The internal dose makes the poison: higher internalization of polystyrene particles induce increased perturbation of macrophages. Front. Immunol. 14, 1092743 (2023). AtlasPubMed
- 3.↑ Catarino, A. I., Frutos, A. & Henry, T. B. Use of fluorescent-labelled nanoplastics (NPs) to demonstrate NP absorption is inconclusive without adequate controls. Sci. Total Environ. 670, 915-920 (2019). AtlasPubMed
- 4.↑ Feng, X., Liu, S. & Xu, H. Not just protons: Chloride also activates lysosomal acidic hydrolases. J. Cell Biol. 222, e202305007 (2023). PubMed
- 5.↑ Rejman, J., Oberle, V., Zuhorn, I. S. & Hoekstra, D. Size-dependent internalization of particles via the pathways of clathrin- and caveolae-mediated endocytosis. Biochem. J. 377, 159-169 (2004). PubMed
- 6.↑ Lunov, O. et al. Amino-Functionalized Polystyrene Nanoparticles Activate the NLRP3 Inflammasome in Human Macrophages. ACS Nano 5, 9648-9657 (2011). PubMed
- 7.↑ Zeng, J., Shirihai, O. S. & Grinstaff, M. W. Modulating lysosomal pH: a molecular and nanoscale materials design perspective. JoLS, J. Life Sci. 2, 25-37 (2020). PubMed
- 8.↑ Merkley, S. D. et al. Polystyrene microplastics induce an immunometabolic active state in macrophages. Cell Biol. Toxicol. 38, 31-41 (2022). AtlasPubMed
- 9. Moosavi, S. S., Ospina, H. A. & Descoteaux, A. Polystyrene Nanoplastics Increase Macrophage Bactericidal Activity Through a Mechanism Involving Reactive Oxygen Species and Itaconate. Nanomaterials 16, 105 (2026). PubMed
- 10.↑ Han, S. et al. Endosomal sorting results in a selective separation of the protein corona from nanoparticles. Nat. Commun. 14, 295 (2023). PubMed
- 11.↑ Kustra, A., Maliszewska-Olejniczak, K., Sekrecka-Belniak, A., Kulawiak, B. & Bednarczyk, P. Polystyrene Nanoplastics in Human Gastrointestinal Models—Cellular and Molecular Mechanisms of Toxicity. Int. J. Mol. Sci. 26, 11738 (2025). AtlasPubMed
- 12.↑ Bochicchio, D., Panizon, E., Monticelli, L. & Rossi, G. Interaction of hydrophobic polymers with model lipid bilayers. Sci. Rep. 7, 6357 (2017). PubMed
- 13.↑ Ramsperger, A. F. R. M. et al. Environmental exposure enhances the internalization of microplastic particles into cells. Sci. Adv. 6, eabd1211 (2020). AtlasPubMed
- 14.↑ Mindell, J. A. Lysosomal Acidification Mechanisms. Annu. Rev. Physiol. 74, 69–86 (2012). PubMed
- 15.↑ Deng, J. et al. Microplastics released from food containers can suppress lysosomal activity in mouse macrophages. J. Hazard. Mater. 435, 128980 (2022). AtlasPubMed
- 16.↑ Liu, Z. et al. Lysosomal dependent transcytosis of polystyrene nanoplastics within macrophages. Food Chem. Toxicol. 206, 115771 (2025). AtlasPubMed
- 17. Domenech, J. et al. Long-Term Effects of Polystyrene Nanoplastics in Human Intestinal Caco-2 Cells. Biomolecules 11, 1442 (2021).
- 18.↑ Moreno-Echeverri, A. M. et al. Pitfalls in methods to study colocalization of nanoparticles in mouse macrophage lysosomes. J. Nanobiotechnology 20, 464 (2022).
- 19. Choi, D. S. et al. Label-Free Live-Cell Imaging of Internalized Microplastics and Cytoplasmic Organelles with Multicolor CARS Microscopy. Environ. Sci. Technol. 56, 3045–3055 (2022). AtlasPubMed
- 20.↑ Ribeiro, F., Duarte, A. C. & Costa, J. P. da. Staining methodologies for microplastics screening. TrAC Trends Anal. Chem. 172, 117555 (2024).
- 21.↑ Kaur, J. et al. Label-free detection of polystyrene nanoparticles in Daphnia magna using Raman confocal mapping. Nanoscale Adv. 5, 3453–3462 (2023). PubMed
- 22.↑ Jin, J., Arciszewski, J., Auclair, K. & Jia, Z. Enzymatic polyethylene biorecycling: Confronting challenges and shaping the future. J. Hazard. Mater. 460, 132449 (2023). PubMed
- 23.↑ Temporiti, M. E. E., Nicola, L., Nielsen, E. & Tosi, S. Fungal Enzymes Involved in Plastics Biodegradation. Microorganisms 10, 1180 (2022). PubMed
- 24.↑ Pujol, M. et al. Harnessing Colloidal Dispersion for Laccase-Driven Enzymatic Depolymerization of Polystyrene. Angew. Chem. Int. Ed. 65, e13937 (2026). PubMed
- 25.↑ Hou, L. & Majumder, E. L.-W. Potential for and Distribution of Enzymatic Biodegradation of Polystyrene by Environmental Microorganisms. Materials 14, 503 (2021). PubMed
- 26.↑ Jiang, Y. et al. Soil microbes in the Tibetan Plateau degrade polyvinyl chloride and harbor novel dehalogenase SerB. Environ. Int. 202, 109708 (2025). PubMed
- 27.↑ Yoshida, S. et al. A bacterium that degrades and assimilates poly(ethylene terephthalate). Science 351, 1196–1199 (2016). PubMed
- 28.↑ Tournier, V. et al. An engineered PET depolymerase to break down and recycle plastic bottles. Nature 580, 216–219 (2020). PubMed
- 29.↑ Zhang, Z. et al. Polyvinyl chloride degradation by a bacterium isolated from the gut of insect larvae. Nat. Commun. 13, 5360 (2022). PubMed
- 30. Manabe, S. et al. Treatment of polyethylene microplastics degraded by ultraviolet light irradiation causes lysosome-deregulated cell death. Sci. Rep. 14, 24008 (2024). AtlasPubMed
- 31.↑ Tavakolpournegari, A. et al. Harmful effects of true-to-life nanoplastics derived from PET water bottles in human alveolar macrophages. Environ. Pollut. 348, 123823 (2024). AtlasPubMed
- 32.↑ Ahn, J. et al. Polystyrene Nanoplastics Exacerbate HFD-induced MASLD by Reducing Cathepsin Activity and Triggering Large Vacuole Formation via Impaired Lysosomal Acidification. Int. J. Biol. Sci. 21, 3867–3885 (2025). AtlasPubMed
- 33. Hafeman, A. E. et al. Characterization of the degradation mechanisms of lysine-derived aliphatic poly(ester urethane) scaffolds. Biomaterials 32, 419–429 (2011). PubMed
- 34. Baltazar, G. C. et al. Acidic Nanoparticles Are Trafficked to Lysosomes and Restore an Acidic Lysosomal pH and Degradative Function to Compromised ARPE-19 Cells. PLoS ONE 7, e49635 (2012). PubMed
- 35.↑ Malhotra, M., Surnar, B. & Jayakannan, M. Polymer Topology Driven Enzymatic Biodegradation in Polycaprolactone Block and Random Copolymer Architectures for Drug Delivery to Cancer Cells. Macromolecules 49, 8098–8112 (2016).
- 36.↑ Sahu, K. L. et al. Advancements in polycaprolactone nanoparticles for targeted drug delivery in breast cancer treatment: Strategies and challenges. Next Nanotechnol. 8, 100206 (2025).
- 37.↑ Zhu, Z. et al. Polystyrene nanoplastics induce apoptosis of human kidney proximal tubular epithelial cells via oxidative stress and MAPK signaling pathways. Environ. Sci. Pollut. Res. 30, 110579–110589 (2023). AtlasPubMed
- 38.↑ Lismont, C., Revenco, I. & Fransen, M. Peroxisomal Hydrogen Peroxide Metabolism and Signaling in Health and Disease. Int. J. Mol. Sci. 20, 3673 (2019). PubMed
- 39.↑ Tan, Y. et al. Intrinsic Peroxidase-like Activity of Polystyrene Nanoplastics Mediates Oxidative Stress. Environ. Sci. Technol. 58, 15475–15485 (2024). PubMed
- 40.↑ Hwang, J. et al. Potential toxicity of polystyrene microplastic particles. Sci. Rep. 10, 7391 (2020). AtlasPubMed
- 41.↑ Milosevic, A. M. et al. Assessing the Stability of Fluorescently Encoded Nanoparticles in Lysosomes by Using Complementary Methods. Angew. Chem. Int. Ed. 56, 13382–13386 (2017). PubMed
- 42.↑ Ding, R. et al. Size-dependent toxicity of polystyrene microplastics on the gastrointestinal tract: Oxidative stress related-DNA damage and potential carcinogenicity. Sci. Total Environ. 912, 169514 (2024). AtlasPubMed
- 43.↑ Lambert, S. & Wagner, M. Formation of microscopic particles during the degradation of different polymers. Chemosphere 161, 510–517 (2016). AtlasPubMed
- 44.↑ Araujo, C. F., Nolasco, M. M., Ribeiro, A. M. P. & Ribeiro-Claro, P. J. A. Identification of microplastics using Raman spectroscopy: Latest developments and future prospects. Water Res. 142, 426–440 (2018). AtlasPubMed
Sign in to start a discussion.