The gastric imprint of microplastic exposure
How microplastics interact with the gut barrier, microbiome, and metabolic signaling
Plastic pollution is now recognized not only as an environmental problem but also as a biological one. Microplastics and nanoplastics have been detected in human stool, blood, lung tissue, placental tissue, and even arterial plaques [1-8]. These discoveries confirm that modern plastic exposure does not stop at the environment. It enters the body and interacts with living systems.
One of the first and most important biological environments that ingested microplastics encountered is the intestinal tract. The gut is not simply a digestive tube. It is a complex ecological interface between the outside world and the internal physiology of the body. Its epithelial barrier regulates nutrient absorption, prevents microbial invasion, and communicates with immune and metabolic systems throughout the body. Surrounding and supporting this barrier is the gut microbiome, a dense ecosystem of trillions of microorganisms that influence digestion, immune function, metabolic signaling, and inflammation.
Emerging experimental evidence suggests that microplastics are not biologically inert within this system. Instead, they interact with mucus, epithelial cells, and microbial communities in ways that can alter the structural and functional stability of the gut.
Across multiple experimental systems, several recurring biological patterns have been observed. Microplastic exposure has been associated with thinning of the intestinal mucus layer, reductions in tight junction punctions that maintain barrier integrity, and shifts in microbial populations away from beneficial taxa. These changes frequently coincide with reductions in short chain fatty acids such as butyrate, a key metabolite required for maintaining epithelial health and regulating inflammation.
Taken together, these findings suggest that microplastics may influence the gut ecosystem at several levels simultaneously. They appear capable of affecting physical barrier structures, microbial composition, and metabolic signaling pathways.
While much of the research remains in early stages, the consistency of these patterns across models suggest that microplastics deserve serious consideration as a biological stressor within the gastrointestinal environment.
Entry: microplastics in the digestive system
Human exposure to microplastics occurs through multiple everyday pathways. Food packaging materials, synthetic textiles, tire wear particles, plastic bottles, household dust, and environmental contamination all contribute to a constant background presence of microplastic fragments in modern life.
These particles enter the body primarily through ingestion. Microplastics have been detected in drinking water, seafood, salt, fruits, vegetables, and other foods [9-22]. Inhaled airborne particles can also become trapped in airway mucus and subsequently swallowed, adding another route of entry into the digestive tract.

Once ingested, these particles travel through the gastrointestinal system. Historically, it was often assumed that most microplastics would simply pass through the digestive tract without meaningful biological interaction. However, growing evidence suggests that the situation is more complex.
Studies have demonstrated that microplastics can interact physically with intestinal mucus and epithelial surfaces. Some particles adhere to the mucus layer that coats the intestinal lining. Others can come into direct contact with epithelial cells or become incorporated into microbial biofilms within the gut environment [23-32].
Particle size appears to play a significant role in these interactions [33-35]. Larger microplastics may remain largely within the intestinal lumen, while smaller particles, particularly nanoplastics, have greater potential to interact with cellular surfaces or penetrate biological barriers.
Evidence of regular ingestion is now well established. Multiple studies have identified microplastics in human stool samples [5], confirming that plastic particles routinely pass through the gastrointestinal tract. However, laboratory studies indicate that even transient exposure within the gut may be sufficient to trigger biological responses.
Rather than behaving as passive particles, microplastics appear capable of interacting with the complex ecosystem of the gut in ways that may influence both microbial composition and barrier integrity.

Damage to the gut barrier
The intestinal barrier is one of the most remarkable structures in human physiology. Although it is only a single layer of epithelial cells thick, it performs a highly complex task. It must selectively absorb nutrients, electrolytes, and water from digested food while simultaneously preventing harmful bacteria, toxins, and inflammatory molecules from entering the blood stream. Simply put, allow the Good in and keep the Bad out.
This balance is maintained through a coordinated system of structural and biological defenses. A protective mucus layer forms the first physical barrier between microbes and epithelial cells. Beneath this mucus, epithelial cells are connected by tight junction proteins that regulate the permeability of the barrier. Surrounding these structures is the gut microbiome, which helps regulate immune signaling and maintains the stability of the barrier.
Microplastic exposure appears capable of disturbing each of these protective systems.
Experimental studies have observed structural changes in the intestinal barrier following microplastic exposure. These changes include thinning of the mucus layer, reductions in tight junction proteins, and increased permeability of the intestinal lining. When these protective features weaken, the barrier becomes less effective at preventing microbial components and inflammatory molecules from entering circulation.
In one study [27], exposure to polystyrene microplastics (PSMPs) produced clear, dose dependent damage to the jejunum. Histological analysis showed increasing intestinal injury as concentrations rose, beginning with mild inflammatory cell infiltration at 20 and 100 ppb and progressing to villus shedding at 200 ppb. At the highest exposure level (500 ppb), severe inflammatory infiltration, mucosal rupture, and glandular damage were observed. Microplastic exposure also reduced the number of goblet cells and significantly decreased mucus secretion, with reductions of roughly 45 to 50 percent in the 200 and 500 ppb groups compared with controls. Biomarkers of intestinal barrier injury, including DAO and iFABP, increased in serum at higher exposure levels, while secretory IgA in the jejunum declined, indicating impaired mucosal immunity. Together, these findings suggest that PSMP exposure disrupts intestinal barrier
The result can be a state sometimes described as increased intestinal permeability. Under these conditions, molecules that are normally contained within the gut lumen may begin to cross the epithelial barrier and interact with immune cells in underlying tissues.
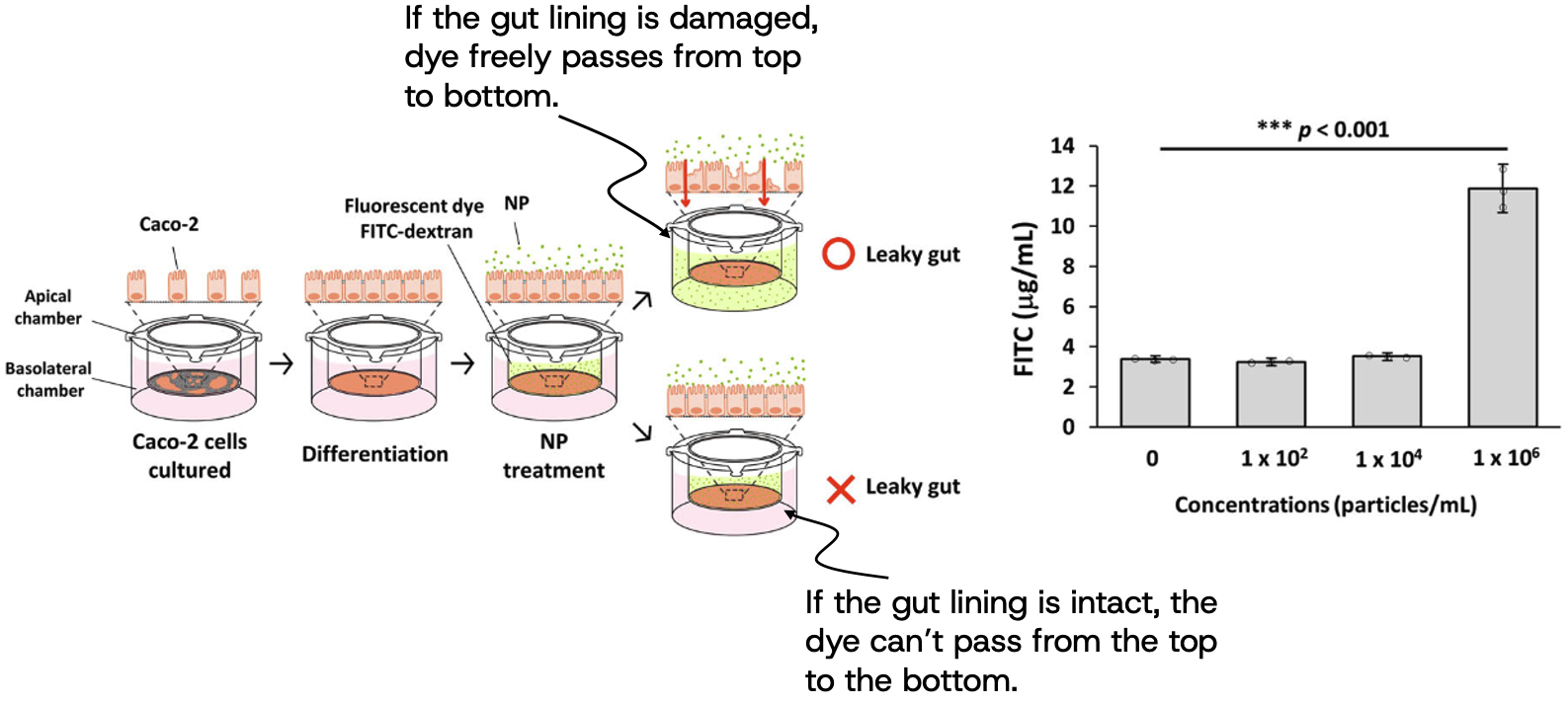
A clear illustration of this barrier disruption appears in a recent study published in Nature Communications [36]. In this work, researchers exposed intestinal cell models and mice to polystyrene nanoplastics and observed direct impairment of the gut barrier. Nanoplastic particles were shown to enter intestinal epithelial cells and significantly reduce the expression of key tight junction proteins, including ZO-1 and occludin. Figure 2 of the study provides a visual record of this process. Microscopy images show nanoplastic particles accumulating within enterocyte like Caco 2 cells, followed by visible disruption of the tight junction network that normally seals intestinal cells together.
They further demonstrated barrier damage using a gut on a chip model. As nanoparticle concentrations increased, the expression of tight junction proteins declined and the permeability of the epithelial layer rose, allowing tracer molecules to pass through the barrier more easily. This effect became most pronounced at approximately 106 particles per milliliter, shown in the bar chart on the right.
But what does 106 particles per milliliter actually represent? For perspective, one million particles of 100 nm polystyrene correspond to only about 0.55 nanograms of plastic per milliliter. A single grain of table salt weighs roughly 60 million nanograms. In other words, each milliliter of the experimental solution contained around 100 million times less plastic mass than a grain of salt, even though it still contained one million individual particles.
While the long term implications of these changes remain under investigation, the disruption of barrier integrity is widely recognized as an important factor in inflammatory and metabolic disorders.
Mucus layer disruption
The mucus layer represents the first line of defense within the intestinal barrier. Produced by specialized goblet cells embedded in the intestinal epithelium, this viscous gel forms a physical shield that separates trillions of gut microbes from direct contact with epithelial cells.

This separation is critical. The microbiome contains both beneficial organisms and potentially harmful species. By maintaining distance between microbes and intestinal tissue, the mucus layer allows beneficial microbial activity to occur while limiting inflammatory interactions with host cells.
Research indicates that microplastic exposure may compromise this protective layer.
Several experimental studies have found reductions in mucus production following exposure to microplastic particles [37-42]. These changes appear to involve both structural and genetic mechanisms. Goblet cell numbers may decrease, and the expression of mucin genes responsible for mucus production can be reduced.
Proteins such as MUC1, MUC2, and MUC3 are central components of intestinal mucus. Reductions in the expression of these proteins lead to a thinner and less protective mucus layer.
When this protective barrier is weakened, bacteria and microbial metabolites can approach epithelial cells more closely. This increased proximity raises the likelihood of immune activation and inflammatory signaling within the intestinal wall.
The mucus layer also interacts closely with certain beneficial microbes. Akkermansia muciniphila, for example, helps maintain healthy mucus turnover and barrier function. Microplastic exposure has been associated with reductions in this bacterium, suggesting a reinforcing cycle in which microbial shifts and mucus depletion may amplify one another.
Tight Junction Disruption and Increased Permeability
Beneath the mucus layer lies the epithelial cell layer that forms the core of the intestinal barrier. These epithelial cells are connected to one another through structures known as tight junctions.
Tight junction proteins function as molecular seals between adjacent cells. They regulate which molecules can pass between cells and enter underlying tissues.
Key proteins involved in this system include claudin 1, occludin, and ZO 1. Together, they form a dynamic gate that maintains the selective permeability of the intestinal barrier.
Experimental studies suggest that microplastic exposure can reduce the expression of these proteins. When tight junction protein levels decline, the barrier becomes more permeable.

Increased permeability allows molecules that normally remain confined within the intestinal lumen to cross the epithelial layer. Among the most biologically significant of these molecules is lipopolysaccharide, a component of the outer membrane of certain bacteria.
Lipopolysaccharide is a powerful immune stimulant. Even small amounts entering the bloodstream can trigger systemic inflammatory signaling.
Chronic low level exposure to lipopolysaccharide has been linked to metabolic disorders, insulin resistance, and inflammatory diseases.
In laboratory models, microplastic exposure has been associated with elevated inflammatory cytokines including TNF alpha, IL 1 beta, and IL 6. At the same time, levels of anti inflammatory cytokines such as IL 10 may decrease.
These shifts suggest that barrier disruption may initiate broader immune responses beyond the gut itself.
Microbiome Disruption
The gut microbiome is a complex ecosystem composed of thousands of microbial species. These microbes perform essential functions in digestion, vitamin synthesis, immune education, and metabolic regulation.
A healthy microbiome maintains a dynamic balance between microbial populations. This balance helps prevent the overgrowth of harmful organisms while supporting the beneficial functions of symbiotic bacteria.
Microplastic exposure has repeatedly been associated with disruptions to this microbial equilibrium [43-121].
Studies across multiple animal models have documented reductions in beneficial microbial groups such as Lactobacillus, Bifidobacterium, and Akkermansia. These organisms play important roles in maintaining gut barrier function and regulating immune responses.
At the same time, certain opportunistic or inflammatory species appear to increase in abundance following microplastic exposure.
Examples include Desulfovibrio, Escherichia Shigella, and several Clostridium related taxa. Some of these microbes produce metabolites that can damage epithelial cells or promote inflammation within the gut.
This shift toward a more inflammatory microbial community is commonly referred to as dysbiosis.
Dysbiosis does not simply reflect changes in microbial composition. It often corresponds with alterations in microbial metabolism and signaling. As microbial populations change, the biochemical products they produce also shift.
These metabolic changes can influence host physiology, particularly in relation to immune signaling and metabolic regulation.
Disruption of Short Chain Fatty Acid Metabolism
Among the most important metabolic products generated by the gut microbiome are short chain fatty acids. These molecules are produced when beneficial bacteria ferment dietary fibers that reach the colon.
The primary short chain fatty acids are acetate, propionate, and butyrate.
Butyrate holds particular importance for intestinal health. It serves as the primary energy source for colonocytes, the epithelial cells lining the colon. Adequate butyrate supply supports epithelial repair, maintains tight junction integrity, and helps regulate inflammatory signaling pathways.
Butyrate also plays roles beyond the intestine. It influences immune cell differentiation, metabolic regulation, and even communication between the gut and the brain.
Microplastic exposure has been associated with reductions in bacteria responsible for butyrate production [39,65,120,122-123]. These include members of the genera Faecalibacterium, Roseburia, and Butyricicoccaceae.
When these microbial populations decline, butyrate production decreases.
Reduced butyrate availability can weaken epithelial cells, making the intestinal barrier more vulnerable to injury and inflammation. In parallel, changes in acetate and propionate levels may alter metabolic signaling related to appetite regulation and hepatic glucose production.
These metabolic changes may represent one of the key pathways through which microplastic induced dysbiosis could influence systemic physiology.
Cellular Stress and Systemic Effects
At the cellular level, microplastics appear capable of inducing oxidative stress and disrupting mitochondrial function [124-130].
Mitochondria act as the energy producing structures within cells. When mitochondrial activity is impaired, cells may produce excess reactive oxygen species while generating less usable energy.
This imbalance can damage cellular structures and activate inflammatory pathways.
Experimental models have observed mitochondrial dysfunction in intestinal cells exposed to microplastic particles. These effects are often accompanied by increased oxidative stress markers and elevated inflammatory cytokine expression.
When viewed at the level of whole organs, these cellular changes may contribute to broader metabolic disturbances.
Animal studies have reported liver inflammation, altered lipid metabolism, and abnormal fat accumulation following chronic microplastic exposure.
Although human research is still emerging, these findings suggest that intestinal interactions with microplastics could potentially influence systemic metabolic pathways.
A Converging Pattern of Evidence
Across experimental systems, a consistent biological pattern appears to be emerging.

Microplastic exposure is associated with weakening of the gut’s structural defenses, disruption of beneficial microbial populations, and reductions in microbial metabolites that normally support intestinal health.
At the same time, inflammatory signaling pathways appear to become more active.
These changes create a physiological environment that is more permeable, more metabolically stressed, and more prone to inflammatory signaling.
The gut is one of the body’s most important interfaces with environmental exposures. When disturbances occur within this system, their effects can extend beyond digestion to influence immune regulation, metabolic health, and systemic inflammation.
Although many questions remain unanswered, the existing evidence suggests that microplastics are capable of interacting with biological systems in ways that merit continued scientific investigation.
References
- 1.↑ Leslie, H. A. et al. Discovery and quantification of plastic particle pollution in human blood. Environ. Int. 163, 107199 (2022). PubMed
- 2. Ragusa, A. et al. Plasticenta: First evidence of microplastics in human placenta. Environ. Int. 146, 106274 (2021). AtlasPubMed
- 3. Saraluck, A. et al. Detection of Microplastics in Human Breast Milk and Its Association with Changes in Human Milk Bacterial Microbiota. J. Clin. Med. 13, 4029 (2024). AtlasPubMed
- 4. Ragusa, A. et al. Raman Microspectroscopy Detection and Characterisation of Microplastics in Human Breastmilk. Polymers 14, 2700 (2022). AtlasPubMed
- 5.↑ Schwabl, P. et al. Detection of Various Microplastics in Human Stool: A Prospective Case Series. Ann. Intern. Med. 171, 453–457 (2019). AtlasPubMed
- 6. Hartmann, C. et al. Assessment of microplastics in human stool: A pilot study investigating the potential impact of diet-associated scenarios on oral microplastics exposure. Sci. Total Environ. 951, 175825 (2024). AtlasPubMed
- 7. Marfella, R. et al. Microplastics and Nanoplastics in Atheromas and Cardiovascular Events. N. Engl. J. Med. 390, 900–910 (2024).
- 8.↑ Jenner, L. C. et al. Detection of microplastics in human lung tissue using μFTIR spectroscopy. Sci. Total Environ. 831, 154907 (2022). AtlasPubMed
- 9.↑ Cox, K. D. et al. Human Consumption of Microplastics. Environ. Sci. Technol. 53, 7068–7074 (2019). https://pubmed.ncbi.nlm.nih.gov/31184127/; Cox, K. D. et al. Correction to Human Consumption of Microplastics. Environ. Sci. Technol. 54, 10974–10974 (2020). AtlasPubMed
- 10. Hernandez, L. M. et al. Plastic Teabags Release Billions of Microparticles and Nanoparticles into Tea. Environ. Sci. Technol. 53, 12300–12310 (2019). AtlasPubMed
- 11. Zangmeister, C. D., Radney, J. G., Benkstein, K. D. & Kalanyan, B. Common Single-Use Consumer Plastic Products Release Trillions of Sub-100 nm Nanoparticles per Liter into Water during Normal Use. Environ. Sci. Technol. 56, 5448–5455 (2022). PubMed
- 12. Oßmann, B. E. et al. Small-sized microplastics and pigmented particles in bottled mineral water. Water Res. 141, 307–316 (2018). AtlasPubMed
- 13. Mehta, N. & Kozielska, B. Disposable Plasticware Production as a Source of Microplastics in Settled Dust: Quantification, Characterization, and Exposure Assessment. Atmosphere 17, 282 (2026).
- 14. Chaïb, I., Doyen, P., Merveillie, P., Dehaut, A. & Duflos, G. Microplastic contaminations in a set of beverages sold in France. J. Food Compos. Anal. 144, 107719 (2025).
- 15. Schymanski, D., Goldbeck, C., Humpf, H.-U. & Fürst, P. Analysis of microplastics in water by micro-Raman spectroscopy: Release of plastic particles from different packaging into mineral water. Water Res. 129, 154–162 (2018). AtlasPubMed
- 16. Hussain, K. A. et al. Assessing the Release of Microplastics and Nanoplastics from Plastic Containers and Reusable Food Pouches: Implications for Human Health. Environ. Sci. Technol. 57, 9782–9792 (2023). AtlasPubMed
- 17. Hu, J.-L. et al. Analysis of microplastics released from plastic take-out food containers based on thermal properties and morphology study. Food Addit. Contam.: Part A 40, 305–318 (2023). AtlasPubMed
- 18. Li, D. et al. Microplastic release from the degradation of polypropylene feeding bottles during infant formula preparation. Nat. Food 1, 746–754 (2020). AtlasPubMed
- 19. Yang, D. et al. Microplastic Pollution in Table Salts from China. Environ. Sci. Technol. 49, 13622–13627 (2015). AtlasPubMed
- 20. Karami, A. et al. Microplastic and mesoplastic contamination in canned sardines and sprats. Sci. Total Environ. 612, 1380–1386 (2018). AtlasPubMed
- 21. Karami, A., Golieskardi, A., Ho, Y. B., Larat, V. & Salamatinia, B. Microplastics in eviscerated flesh and excised organs of dried fish. Sci. Rep. 7, 5473 (2017). AtlasPubMed
- 22.↑ Conti, G. O. et al. Micro- and nano-plastics in edible fruit and vegetables. The first diet risks assessment for the general population. Environ. Res. 187, 109677 (2020). AtlasPubMed
- 23.↑ Jin, Y., Lu, L., Tu, W., Luo, T. & Fu, Z. Impacts of polystyrene microplastic on the gut barrier, microbiota and metabolism of mice. Sci. Total Environ. 649, 308–317 (2019). AtlasPubMed
- 24. Lu, L., Wan, Z., Luo, T., Fu, Z. & Jin, Y. Polystyrene microplastics induce gut microbiota dysbiosis and hepatic lipid metabolism disorder in mice. Sci. Total Environ. 631, 449–458 (2018). AtlasPubMed
- 25. Wei, G. et al. Polystyrene microplastics exposure increases the disruption of intestinal barrier integrity and gut microbiota homeostasis during obesity and aging. J. Environ. Sci. (China) 162, 430–443 (2025). AtlasPubMed
- 26. Wang, H., Liu, B. & Zhao, X. New Insights into the Relationship Between Microplastics and Diabetes from the Perspective of the Gut–Liver Axis and Macrophage Regulation. Toxics (2026) doi:10.3390/toxics14030241.
- 27.↑ Li, H. et al. Polystyrene microplastics exposure: Disruption of intestinal barrier integrity and hepatic function in infant mice. Ecotoxicol. Environ. Saf. 288, 117357 (2024). AtlasPubMed
- 28. Sánchez, A. et al. Microplastic-induced alterations in the intestinal environment: Aging as a modulating factor. Toxicology 522, 154427 (2026). AtlasPubMed
- 29. Deng, Y., Zhang, Y., Lemos, B. & Ren, H. Tissue accumulation of microplastics in mice and biomarker responses suggest widespread health risks of exposure. Sci. Rep. 7, 46687 (2017). https://pubmed.ncbi.nlm.nih.gov/28436478/; Tang, B. L. Commentary: Tissue accumulation of microplastics in mice and biomarker responses suggest widespread health risks of exposure. Front. Environ. Sci. 5, 63 (2017). AtlasPubMed
- 30. Sung, M. et al. Exacerbation of polyethylene microplastics in animal models of DSS-induced colitis through damage to intestinal epithelial cell conjunctions. Curr. Res. Toxicol. 8, 100217 (2025). AtlasPubMed
- 31. Agrawal, M. et al. Micro- and nano-plastics, intestinal inflammation, and inflammatory bowel disease: A review of the literature. Sci. Total Environ. 953, 176228 (2024). AtlasPubMed
- 32.↑ Hasegawa, Y. et al. Oral exposure to high concentrations of polystyrene microplastics alters the intestinal environment and metabolic outcomes in mice. Front. Immunol. 15, 1407936 (2024).
- 33.↑ Wright, S. L. & Kelly, F. J. Plastic and Human Health: A Micro Issue? Environ. Sci. Technol. 51, 6634–6647 (2017). AtlasPubMed
- 34. Luo, T. et al. Polystyrene microplastics exacerbate experimental colitis in mice tightly associated with the occurrence of hepatic inflammation. Sci. Total Environ. 844, 156884 (2022). AtlasPubMed
- 35.↑ Wang, Z. et al. Polystyrene microplastics induce potential toxicity through the gut-mammary axis. npj Sci. Food 9, 139 (2025). AtlasPubMed
- 36.↑ Zolotova, N. et al. Influence of Microplastics on Manifestations of Experimental Chronic Colitis. Toxics 13, 701 (2025). AtlasPubMed
- 37.↑ Zolotova, N., Dzhalilova, D., Tsvetkov, I. & Makarova, O. Influence of Microplastics on Morphological Manifestations of Experimental Acute Colitis. Toxics 11, 730 (2023). AtlasPubMed
- 38. Okamura, T. et al. Oral Exposure to Polystyrene Microplastics of Mice on a Normal or High-Fat Diet and Intestinal and Metabolic Outcomes. Environ. Heal. Perspect. 131, 027006 (2023). AtlasPubMed
- 39.↑ Niu, H. et al. Differential Impacts of Environmentally Relevant Microplastics on Gut Barrier Integrity in Mice Fed High-Fat Diet Versus Normal Chow Diet. Metabolites 15, 557 (2025). AtlasPubMed
- 40. Sun, H., Chen, N., Yang, X., Xia, Y. & Wu, D. Effects induced by polyethylene microplastics oral exposure on colon mucin release, inflammation, gut microflora composition and metabolism in mice. Ecotoxicol. Environ. Saf. 220, 112340 (2021). AtlasPubMed
- 41. Gazzar, W. B. E. et al. Melatonin Alleviates Intestinal Barrier Damaging Effects Induced by Polyethylene Microplastics in Albino Rats. Int. J. Mol. Sci. 24, 13619 (2023). AtlasPubMed
- 42.↑ Gan, H.-J. et al. Simulated Microplastic Release from Cutting Boards and Evaluation of Intestinal Inflammation and Gut Microbiota in Mice. Environ. Heal. Perspect. 133, 047004 (2025). AtlasPubMed
- 43.↑ Chen, X. et al. Polyvinyl chloride microplastics induced gut barrier dysfunction, microbiota dysbiosis and metabolism disorder in adult mice. Ecotoxicol. Environ. Saf. 241, 113809 (2022). AtlasPubMed
- 44. Chen, W. et al. Cyanidin-3-O-glucoside impacts fecal discharge of polystyrene microplastics in mice: Potential role of microbiota-derived metabolites. Toxicol. Appl. Pharmacol. 453, 116212 (2022). AtlasPubMed
- 45. Chen, Y. et al. Polystyrene microplastics aggravate radiation-induced intestinal injury in mice. Ecotoxicol. Environ. Saf. 283, 116834 (2024). AtlasPubMed
- 46. Chen, J. et al. Multiomics Reveals Nonphagocytosable Microplastics Induce Colon Inflammatory Injury via Bile Acid-Gut Microbiota Interactions and Barrier Dysfunction. ACS Appl. Mater. Interfaces 17, 44138–44159 (2025). AtlasPubMed
- 47. Chen, D. et al. Microbial diversity and metabolomics analysis of colon contents exposed to cadmium and polystyrene microplastics. Ecotoxicol. Environ. Saf. 290, 117585 (2025). AtlasPubMed
- 48. Chi, J. et al. Metabolic Reprogramming in Gut Microbiota Exposed to Polystyrene Microplastics. Biomedicines 13, 446 (2025). AtlasPubMed
- 49. Choi, Y. S. et al. Dysbiosis of gut microbiota in C57BL/6-Lepem1hwl/Korl mice during microplastics-caused hepatic metabolism disruption. PLOS One 20, e0336627 (2025). AtlasPubMed
- 50. Deng, Y. et al. Microplastics release phthalate esters and cause aggravated adverse effects in the mouse gut. Environ. Int. 143, 105916 (2020). AtlasPubMed
- 51. Gan, H.-J. et al. Simulated Microplastic Release from Cutting Boards and Evaluation of Intestinal Inflammation and Gut Microbiota in Mice. Environ. Heal. Perspect. 133, 047004 (2025). AtlasPubMed
- 52. Gao, B. et al. Size-dependent effects of polystyrene microplastics on gut metagenome and antibiotic resistance in C57BL/6 mice. Ecotoxicol. Environ. Saf. 254, 114737 (2023). AtlasPubMed
- 53. Gao, B. et al. Mixture Effects of Polystyrene Microplastics on the Gut Microbiota in C57BL/6 Mice. ACS Omega 10, 7597–7608 (2025). AtlasPubMed
- 54. Gao, B. et al. Association between microplastics and the functionalities of human gut microbiome. Ecotoxicol. Environ. Saf. 290, 117497 (2025). AtlasPubMed
- 55. Hasegawa, Y. et al. Oral exposure to high concentrations of polystyrene microplastics alters the intestinal environment and metabolic outcomes in mice. Front. Immunol. 15, 1407936 (2024). AtlasPubMed
- 56. Hsu, W.-H. et al. Polystyrene nanoplastics disrupt the intestinal microenvironment by altering bacteria-host interactions through extracellular vesicle-delivered microRNAs. Nat. Commun. 16, 5026 (2025). AtlasPubMed
- 57. Huang, W. et al. Influence of the co-exposure of microplastics and tetrabromobisphenol A on human gut: Simulation in vitro with human cell Caco-2 and gut microbiota. Sci. Total Environ. 778, 146264 (2021). AtlasPubMed
- 58. Huang, H.-J. et al. Polystyrene Microplastics Can Aggravate the Damage of the Intestinal Microenvironment Caused by Okadaic Acid: A Prevalent Algal Toxin. Mar. Drugs 23, 129 (2025). AtlasPubMed
- 59. Hwangbo, H. et al. Polystyrene Accelerates Aging Related-Gut Microbiome Dysbiosis and -Metabolites in Old-Aged Mouse. J. Microbiol. Biotechnol. 35, 1–11 (2025). AtlasPubMed
- 60. Jeong, B. et al. Maternal nanoplastic ingestion induces an increase in offspring body weight through altered lipid species and microbiota. Environ. Int. 185, 108522 (2024). AtlasPubMed
- 61. Jiménez-Arroyo, C. et al. Simulated gastrointestinal digestion of polylactic acid (PLA) biodegradable microplastics and their interaction with the gut microbiota. Sci. Total Environ. 902, 166003 (2023). AtlasPubMed
- 62. Jin, Y., Lu, L., Tu, W., Luo, T. & Fu, Z. Impacts of polystyrene microplastic on the gut barrier, microbiota and metabolism of mice. Sci. Total Environ. 649, 308–317 (2019). AtlasPubMed
- 63. Kaluç, N. et al. Gut-lung microbiota dynamics in mice exposed to Nanoplastics. NanoImpact 36, 100531 (2024). AtlasPubMed
- 64. Ke, D. et al. Occurrence of microplastics and disturbance of gut microbiota: a pilot study of preschool children in Xiamen, China. eBioMedicine 97, 104828 (2023). AtlasPubMed
- 65.↑ Kim, K. J. et al. In vivo exposure of mixed microplastic particles in mice and its impacts on the murine gut microbiome and metabolome. Toxicol. Sci. 209, kfaf145 (2025). AtlasPubMed
- 66. Kuai, Y. et al. Long-term exposure to polystyrene microplastics reduces macrophages and affects the microbiota–gut–brain axis in mice. Toxicology 509, 153951 (2024). AtlasPubMed
- 67. Li, B. et al. Polyethylene microplastics affect the distribution of gut microbiota and inflammation development in mice. Chemosphere 244, 125492 (2020). AtlasPubMed
- 68. Li, H. et al. Polystyrene microplastics exposure: Disruption of intestinal barrier integrity and hepatic function in infant mice. Ecotoxicol. Environ. Saf. 288, 117357 (2024). AtlasPubMed
- 69. Li, Z., Li, Y., Cao, F., Huang, J. & Gao, X. Gut microbiota and metabolic health risks from chronic low-dose microplastic exposure with focus on Desulfovibrio spp. Ecotoxicol. Environ. Saf. 302, 118721 (2025). AtlasPubMed
- 70. Li, Y. et al. Combined exposure to microplastics and tetracycline leads to impaired skeletal development in young mice by the microbiota-gut-bone axis. Ecotoxicol. Environ. Saf. 306, 119308 (2025). AtlasPubMed
- 71. Li, M. et al. Short-term microplastic exposure: A double whammy to lung metabolism and fecal microflora in diabetic SD rats. Ecotoxicol. Environ. Saf. 297, 118229 (2025). AtlasPubMed
- 72. Li, X., Jing, K., Song, P. & Yu, J. Aged polystyrene microplastics exacerbate cadmium-induced hepatotoxicity in zebrafish through gut-liver axis metabolic dysregulation. Environ. Chem. Ecotoxicol. 7, 859–871 (2025).
- 73. Liang, X., Wang, Y., Andrikopoulos, N., Ke, P. C. & Li, Y. Dysfunctional digestive tract highlights the metabolic hallmarks of nanoplastic-exacerbated Parkinson’s pathology. npj Park.’s Dis. 11, 300 (2025). PubMed
- 74. Lu, L., Wan, Z., Luo, T., Fu, Z. & Jin, Y. Polystyrene microplastics induce gut microbiota dysbiosis and hepatic lipid metabolism disorder in mice. Sci. Total Environ. 631, 449–458 (2018). AtlasPubMed
- 75. Lu, T. et al. Chronic exposure to polyethylene terephthalate microplastics induces gut microbiota dysbiosis and disordered hepatic lipid metabolism in mice. Ecotoxicol. Environ. Saf. 298, 118330 (2025). AtlasPubMed
- 76. Marana, M. H. et al. Plastic nanoparticles cause mild inflammation, disrupt metabolic pathways, change the gut microbiota and affect reproduction in zebrafish: A full generation multi-omics study. J. Hazard. Mater. 424, 127705 (2022). AtlasPubMed
- 77. Nissen, L. et al. Single exposure of food-derived polyethylene and polystyrene microplastics profoundly affects gut microbiome in an in vitro colon model. Environ. Int. 190, 108884 (2024). AtlasPubMed
- 78. Nugrahapraja, H. et al. Effects of Microplastic on Human Gut Microbiome: Detection of Plastic-Degrading Genes in Human Gut Exposed to Microplastics—Preliminary Study. Environments 9, 140 (2022).
- 79. Qiao, J. et al. Perturbation of gut microbiota plays an important role in micro/nanoplastics-induced gut barrier dysfunction. Nanoscale 13, 8806–8816 (2021). AtlasPubMed
- 80. Rehman, A. et al. Impacts of polystyrene nanoplastics on zebrafish gut microbiota and mechanistic insights. Ecotoxicol. Environ. Saf. 299, 118332 (2025). AtlasPubMed
- 81. Shen, W. et al. Sex-Specific Effects of Polystyrene Microplastic and Lead(II) Co-Exposure on the Gut Microbiome and Fecal Metabolome in C57BL/6 Mice. Metabolites 14, 189 (2024). AtlasPubMed
- 82. Su, Q.-L. et al. The impact of microplastics polystyrene on the microscopic structure of mouse intestine, tight junction genes and gut microbiota. PLOS ONE 19, e0304686 (2024). AtlasPubMed
- 83. Sun, H., Chen, N., Yang, X., Xia, Y. & Wu, D. Effects induced by polyethylene microplastics oral exposure on colon mucin release, inflammation, gut microflora composition and metabolism in mice. Ecotoxicol. Environ. Saf. 220, 112340 (2021). AtlasPubMed
- 84. Sun, X. et al. Polyethylene terephthalate microplastics affect gut microbiota distribution and intestinal damage in mice. Ecotoxicol. Environ. Saf. 294, 118119 (2025). AtlasPubMed
- 85. Tamargo, A. et al. PET microplastics affect human gut microbiota communities during simulated gastrointestinal digestion, first evidence of plausible polymer biodegradation during human digestion. Sci. Rep. 12, 528 (2022). AtlasPubMed
- 86. Tilves, C. et al. Associations of Plastic Bottle Exposure with Infant Growth, Fecal Microbiota, and Short-Chain Fatty Acids. Microorganisms 11, 2924 (2023). PubMed
- 87. Tu, P. et al. Deciphering Gut Microbiome Responses upon Microplastic Exposure via Integrating Metagenomics and Activity-Based Metabolomics. Metabolites 13, 530 (2023). AtlasPubMed
- 88. Wang, Z. et al. Transfer toxicity of polystyrene microplastics in vivo: Multi-organ crosstalk. Environ. Int. 202, 109604 (2025). AtlasPubMed
- 89. Wang, S. et al. The influence of microplastics on hypertension-associated cardiovascular injury via the modulation of gut microbiota. Environ. Pollut. 368, 125760 (2025). AtlasPubMed
- 90. Wang, Z. et al. Polystyrene microplastics induce potential toxicity through the gut-mammary axis. npj Sci. Food 9, 139 (2025). AtlasPubMed
- 91. Wei, G. et al. Low-dose polystyrene microplastics exposure increases susceptibility to obesity-induced MASLD via disrupting intestinal barrier integrity and gut microbiota homeostasis. Ecotoxicol. Environ. Saf. 299, 118310 (2025). AtlasPubMed
- 92. Xia, Y. et al. Effects of microplastics and tetracycline induced intestinal damage, intestinal microbiota dysbiosis, and antibiotic resistome: metagenomic analysis in young mice. Environ. Int. 199, 109512 (2025). AtlasPubMed
- 93. Xie, L. et al. Intestinal flora variation reflects the short-term damage of microplastic to the intestinal tract in mice. Ecotoxicol. Environ. Saf. 246, 114194 (2022). AtlasPubMed
- 94. Xu, M. et al. Impact of Microplastic Exposure on Blood Glucose Levels and Gut Microbiota: Differential Effects under Normal or High-Fat Diet Conditions. Metabolites 14, 504 (2024). AtlasPubMed
- 95. Yang, Y.-K.-X. et al. Effects of Polyvinyl Chloride Microplastics on the Reproductive System, Intestinal Structure, and Microflora in Male and Female Mice. Vet. Sci. 11, 488 (2024). AtlasPubMed
- 96. Zhai, Z., Yang, Y., Chen, S. & Wu, Z. Long-Term Exposure to Polystyrene Microspheres and High-Fat Diet–Induced Obesity in Mice: Evaluating a Role for Microbiota Dysbiosis. Environ. Heal. Perspect. 132, 097002 (2024). AtlasPubMed
- 97. Zhang, Z. et al. Continuous oral exposure to micro- and nanoplastics induced gut microbiota dysbiosis, intestinal barrier and immune dysfunction in adult mice. Environ. Int. 182, 108353 (2023). AtlasPubMed
- 98. Zhou, Y. et al. Gut microbiota combined with metabolome dissects long-term nanoplastics exposure-induced disturbed spermatogenesis. Ecotoxicol. Environ. Saf. 267, 115626 (2023). AtlasPubMed
- 99. Agrawal, M. et al. Micro- and nano-plastics, intestinal inflammation, and inflammatory bowel disease: A review of the literature. Sci. Total Environ. 953, 176228 (2024). AtlasPubMed
- 100. Ali, N. et al. The potential impacts of micro-and-nano plastics on various organ systems in humans. eBioMedicine 99, 104901 (2024). AtlasPubMed
- 101. Bora, S. S. et al. Microplastics and human health: unveiling the gut microbiome disruption and chronic disease risks. Front. Cell. Infect. Microbiol. 14, 1492759 (2024). AtlasPubMed
- 102. Chen, X. et al. Adverse health effects of emerging contaminants on inflammatory bowel disease. Front. Public Heal. 11, 1140786 (2023). AtlasPubMed
- 103. Covello, C., Vincenzo, F. D., Cammarota, G. & Pizzoferrato, M. Micro(nano)plastics and Their Potential Impact on Human Gut Health: A Narrative Review. Curr. Issues Mol. Biol. 46, 2658–2677 (2024). AtlasPubMed
- 104. Demarquoy, J. Microplastics and microbiota: Unraveling the hidden environmental challenge. World J. Gastroenterol. 30, 2191–2194 (2024). AtlasPubMed
- 105. Duan, J., Liu, C., Bai, X., Zhao, X. & Jiang, T. Global trends and hotspots of gastrointestinal microbiome and toxicity based on bibliometrics. Front. Microbiol. 14, 1231372 (2023). AtlasPubMed
- 106. Eichinger, J., Tretola, M., Seifert, J. & Brugger, D. Review: interactions between microplastics and the gastrointestinal microbiome. Ital. J. Anim. Sci. 23, 1044–1056 (2024).
- 107. Fournier, E. et al. Microplastics in the human digestive environment: A focus on the potential and challenges facing in vitro gut model development. J. Hazard. Mater. 415, 125632 (2021). AtlasPubMed
- 108. Garrido-Romero, M. et al. Relevance of gut microbiome research in food safety assessment. Gut Microbes 16, 2410476 (2024). AtlasPubMed
- 109. Hirt, N. & Body-Malapel, M. Immunotoxicity and intestinal effects of nano- and microplastics: a review of the literature. Part. Fibre Toxicol. 17, 57 (2020). AtlasPubMed
- 110. Huang, Z., Weng, Y., Shen, Q., Zhao, Y. & Jin, Y. Microplastic: A potential threat to human and animal health by interfering with the intestinal barrier function and changing the intestinal microenvironment. Sci. Total Environ. 785, 147365 (2021). AtlasPubMed
- 111. Jiménez-Arroyo, C., Tamargo, A., Molinero, N. & Moreno-Arribas, M. V. The gut microbiota, a key to understanding the health implications of micro(nano)plastics and their biodegradation. Microb. Biotechnol. 16, 34–53 (2023). PubMed
- 112. Mauliasari, I. R. et al. Benzo(a)pyrene and Gut Microbiome Crosstalk: Health Risk Implications. Toxics 12, 938 (2024). AtlasPubMed
- 113. Mishra, S. K. et al. Microplastics as emerging carcinogens: from environmental pollutants to oncogenic drivers. Mol. Cancer 24, 248 (2025). AtlasPubMed
- 114. Osman, A. I. et al. Microplastic sources, formation, toxicity and remediation: a review. Environ. Chem. Lett. 21, 2129–2169 (2023). AtlasPubMed
- 115. Pacher-Deutsch, C. et al. The microplastic-crisis: Role of bacteria in fighting microplastic-effects in the digestive system. Environ. Pollut. 366, 125437 (2025). AtlasPubMed
- 116. Pan, I. & Umapathy, S. Probiotics an emerging therapeutic approach towards gut-brain-axis oriented chronic health issues induced by microplastics: A comprehensive review. Heliyon 10, e32004 (2024). AtlasPubMed
- 117. Romeo, M. et al. Exploring the Classic and Novel Pathogenetic Insights of Plastic Exposure in the Genesis and Progression of Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD). Livers 5, 21 (2025).
- 118. Sinha, P., Saini, V., Varshney, N., Pandey, R. K. & Jha, H. C. The infiltration of microplastics in human systems: Gastrointestinal accumulation and pathogenic impacts. Heliyon 11, e42606 (2025). AtlasPubMed
- 119. Souza-Silva, T. G. de et al. Impact of microplastics on the intestinal microbiota: A systematic review of preclinical evidence. Life Sci. 294, 120366 (2022). AtlasPubMed
- 120.↑ Thin, Z. S., Chew, J., Ong, T. Y. Y., Ali, R. A. R. & Gew, L. T. Impact of microplastics on the human gut microbiome: a systematic review of microbial composition, diversity, and metabolic disruptions. BMC Gastroenterol. 25, 583 (2025). AtlasPubMed
- 121.↑ Zolotova, N. et al. Microplastic effects on mouse colon in normal and colitis conditions: A literature review. PeerJ 13, e18880 (2025). AtlasPubMed
- 122.↑ Adhikari, M., Biswas, C., Bera, R. & Pramanick, K. Polystyrene microplastics-induced gut microbiota dysbiosis with altered short chain fatty acid is associated with reproductive physiology and endocrine disorder in female zebrafish. J. Hazard. Mater. 500, 140468 (2025). AtlasPubMed
- 123.↑ Yan, Z. et al. Phthalates released from microplastics inhibit microbial metabolic activity and induce different effects on intestinal luminal and mucosal microbiota. Environ. Pollut. 310, 119884 (2022). AtlasPubMed
- 124.↑ Schirinzi, G. F. et al. Cytotoxic effects of commonly used nanomaterials and microplastics on cerebral and epithelial human cells. Environ. Res. 159, 579–587 (2017). AtlasPubMed
- 125. Kadac-Czapska, K., Ośko, J., Knez, E. & Grembecka, M. Microplastics and Oxidative Stress—Current Problems and Prospects. Antioxidants 13, 579 (2024). AtlasPubMed
- 126. Xu, J.-L., Lin, X., Wang, J. J. & Gowen, A. A. A review of potential human health impacts of micro- and nanoplastics exposure. Sci. Total Environ. 851, 158111 (2022). AtlasPubMed
- 127. Liu, H., Li, H., Yao, X., Yan, X. & Peng, R. Environmental nanoplastics induce mitochondrial dysfunction: A review of cellular mechanisms and associated diseases. Environ. Pollut. 382, 126695 (2025). AtlasPubMed
- 128. Kovacs, K., Bodis, J. & Vass, R. A. Microplastics, Endocrine Disruptors, and Oxidative Stress: Mechanisms and Health Implications. Int. J. Mol. Sci. 27, 399 (2025). AtlasPubMed
- 129. Cheng, W. et al. Polystyrene microplastics induce hepatotoxicity and disrupt lipid metabolism in the liver organoids. Sci. Total Environ. 806, 150328 (2022). AtlasPubMed
- 130.↑ Yin, K. et al. Polystyrene microplastics promote liver inflammation by inducing the formation of macrophages extracellular traps. J. Hazard. Mater. 452, 131236 (2023). AtlasPubMed
Sign in to start a discussion.