APOE genotype and microplastic exposure
A technical deep dive into what the science shows and where the critical gaps remain
Microplastics have now been detected in human blood, lungs, placenta, kidneys, and, perhaps most unsettlingly, in post mortem brain tissue. Some data also suggest those concentrations may be increasing over time. In a 2025 study published in Nature Medicine, researchers reported that brain tissue samples from deceased individuals contained higher concentrations of microplastics than either liver or kidney tissue, with polyethylene as the dominant polymer. They also found that samples collected in 2024 contained measurably more plastic than samples from 2016 [1].
The exact values reported in that study are being actively debated. Measuring microplastics in human tissue is technically difficult, and methods such as pyrolysis-gas chromatography-mass spectrometry (Py-GC-MS) depend heavily on how effectively biological material is removed before analysis. That matters because residual biological material can interfere with the signal. Still, even with those caveats, the broader pattern is difficult to dismiss. The samples were processed using the same general method, and the measured concentrations appeared to rise over time. Set against the larger backdrop of increasing plastic contamination in the environment, that signal deserves careful attention.
The idea that very small plastic particles could reach the brain is also not entirely foreign to science. Researchers have spent years studying nanoparticle based delivery systems, including plastic-based particles, as tools for crossing the blood brain barrier (BBB). That literature makes clear that particle behavior can change substantially depending on size, surface charge, chemistry, and functionalization. In other words, the route is biologically plausible, even if the real world behavior of environmental microplastics and nanoplastics remains far less understood.
What we do not know is just as important. We do not yet know whether these particles cause harm in humans, who may be most vulnerable, what doses matter, or how risk may accumulate across years or decades.
This article focuses on one part of that larger question: whether APOE genotype, and specifically the APOE ε4 allele, may shape a person’s neurological susceptibility to microplastic and nanoplastic exposure.
That is a scientifically legitimate question, and an increasingly important one. APOE ε4 may be the strongest known genetic risk factor for late onset Alzheimer’s disease and is carried by roughly a quarter of the population. Early experimental findings and mechanistic reasoning suggest that APOE ε4 biology may overlap with several of the same pathways microplastics are suspected to disrupt. Whether that overlap translates into meaningful real world harm remains unknown. This piece maps what the science suggests, where the evidence is strongest, and where uncertainty still remains.
Why this topic matters
APOE ε4 does not guarantee Alzheimer’s disease. But it does meaningfully increase risk, by about threefold for people with one copy and up to twelvefold for those with two. That increased risk is not random. It appears to arise through biological pathways that are also highly relevant to microplastic and nanoplastic exposure [2]. These include a more fragile blood brain barrier, higher baseline neuroinflammation, disrupted lipid handling in glial cells, and weaker clearance of amyloid beta proteins.
At the same time, microplastics and nanoplastics do not appear to be biologically inert once they enter living systems. Across animal and in vitro studies, they have been shown to increase oxidative stress, activate immune signaling, disrupt barrier integrity, and in some models, worsen the kinds of protein misfolding and aggregation associated with neurodegenerative disease. The particles reported in human brain tissue also appear to skew very small, often in the nanoscale range, which matters because particles at that size are more capable of interacting directly with cells, membranes, and intracellular machinery than larger fragments.
The more important question is whether genetic background helps determine who is most vulnerable. In that context, APOE ε4 stands out as one of the most biologically coherent places to look. What remains unproven is whether internal microplastic exposure and APOE ε4 biology interact in humans in a way that meaningfully alters neurodegenerative risk.
Biological background
What APOE is and why it matters in the brain
The APOE gene encodes apolipoprotein E, a protein that helps transport cholesterol and other lipids throughout the body. In the brain, this role is especially important. Although the brain accounts for only about 2% of body weight, it contains roughly a quarter of the body’s total cholesterol. ApoE is the brain’s main lipid transport protein, produced primarily by astrocytes and microglia.
There are three common forms, or isoforms, of ApoE: ApoE2, ApoE3, and ApoE4. They differ by just two amino acids, at positions 112 and 158, but those small differences lead to meaningful changes in structure and function. ApoE4 folds into a more compact and less stable shape than ApoE3, sometimes described as a molten globule conformation. That instability appears to make it more vulnerable to cleavage, more likely to accumulate inside cells instead of being secreted normally, and less effective at moving lipids where they need to go [3].
Several downstream effects of APOE ε4 are especially relevant here.
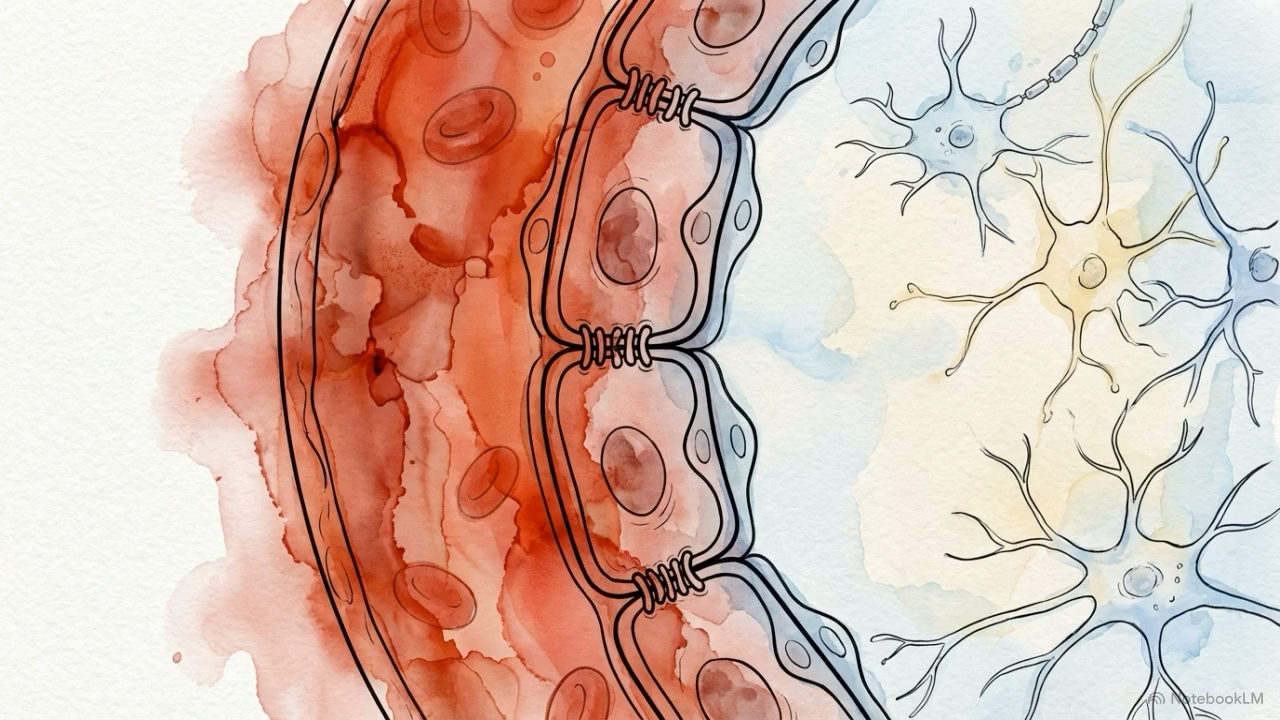
Blood brain barrier fragility. APOE ε4 has been linked to activation of an inflammatory pathway in pericytes involving cyclophilin A, NF-κB, and MMP-9. That cascade can weaken tight junction proteins and make the blood brain barrier more permeable, allowing blood derived molecules to leak more easily into brain tissue [4].
Glial lipid dysregulation. In APOE ε4 carriers, astrocytes and microglia appear more likely to accumulate lipid droplets, recycle cholesterol less efficiently, and shift toward metabolically stressed, more inflammatory states [5].
Heightened neuroinflammatory tone. APOE ε4 microglia tend to sit closer to an activated state. They often produce more inflammatory signals such as TNF-α, IL-1β, and IL-6, and may trigger inflammasome pathways such as NLRP3 more readily in response to stress.
Impaired amyloid beta clearance. ApoE helps clear amyloid beta from the brain’s interstitial fluid. Compared with ApoE3 or ApoE2, ApoE4 appears less effective at that job, which may contribute to greater amyloid accumulation over time [6].
Taken together, these effects help explain why APOE ε4 matters so much in neurodegeneration. It does not act through a single pathway.
What microplastics and nanoplastics are
Microplastics are plastic particles smaller than 5 mm that arise from the breakdown of larger synthetic materials or are produced at small size from the start. Nanoplastics are smaller still, often defined as particles below 1 µm, though definitions vary across the literature. For neurological questions, that smaller fraction matters most, because size strongly influences whether a particle can cross biological barriers.
Submicron and nanoscale particles are more capable of crossing the intestinal lining, entering circulation, and, in experimental models, reaching the brain through processes such as endocytosis and transcytosis. In other words, the smaller the particle, the more biologically mobile it may become.
The polymers most often reported in human tissues include polyethylene, polypropylene, polyvinyl chloride, PET, and polystyrene. In post mortem brain studies, polyethylene appears to predominate. The particles described at that scale also often look less like the smooth spheres commonly used in laboratory experiments and more like irregular shards, flakes, and fragmented debris, which is likely closer to what people encounter in the real world [1].
Human exposure appears to occur mainly through ingestion of food and water, with inhalation as another important route. A more direct nose to brain pathway has also become increasingly plausible. In a 2024 study, researchers detected microplastics in the olfactory bulbs of 8 out of 15 human decedents, while procedural blanks remained negative, strengthening confidence that the signal was real rather than contamination introduced during analysis [7].
That matters because it expands the range of biologically plausible entry points into vulnerable tissue.
Mechanisms
Across the mechanism sections below, the pattern is similar: experimental evidence is often strong, human evidence remains limited, and direct APOE stratified testing is still rare.
Barrier integrity and the blood-brain barrier
The blood brain barrier, or BBB, is one of the brain’s most important protective systems. It is formed by specialized endothelial cells, pericytes, and astrocyte endfeet, and it works as a highly selective filter between the bloodstream and brain tissue. Its integrity depends in part on tight junction proteins such as occludin, claudins, and ZO-1, as well as the health of the surrounding vascular support cells. When that barrier is weakened, inflammatory molecules, blood derived proteins, and potentially foreign particles can gain easier access to the brain.
A growing body of controlled exposure research suggests that microplastics and nanoplastics, especially at very small sizes, can disrupt this barrier through several overlapping mechanisms.
In one mouse study, 50 nm polystyrene nanoplastics increased BBB permeability in a dose dependent manner and were detected in brain tissue after exposure. In a parallel human cerebral microvascular endothelial cell model, those same particles triggered reactive oxygen species, activated NF κB signaling, increased TNF α secretion, reduced occludin expression, and induced endothelial necroptosis, all signs of barrier injury rather than passive contact [8].
Another study using chronic drinking water exposure over 180 days found that polystyrene particles ranging from 0.5 to 10 µm accumulated in the brain, altered tight junction ultrastructure on electron microscopy, increased inflammatory signaling in the hippocampus, and impaired spatial learning in mice [9]. Together, those findings suggest that prolonged exposure may affect not only barrier structure, but also downstream brain function.
Particle behavior also depends on size and surface coating. Kopatz and colleagues showed that nanoplastic passage across the BBB is both size dependent and corona dependent, with smaller particles and cholesterol rich coronas showing greater membrane entry [10].
APOE ε4 enters this picture from a different but highly relevant direction. Independent of any plastic exposure, APOE ε4 is already associated with a more fragile BBB. Bell and colleagues showed that ApoE4 activates cyclophilin A signaling in pericytes, which then drives NF-κB activation, raises MMP-9, degrades tight junction proteins, and allows neurotoxic blood derived material to leak into brain tissue [4]. Human translational work has reinforced that finding. Even cognitively unimpaired APOE ε4 carriers show evidence of hippocampal BBB breakdown, and the cerebrospinal fluid marker sPDGFRβ, a marker of pericyte injury, predicts future cognitive decline independently of other factors [11].
There is also evidence that the ε4 neurovascular environment begins from a more vulnerable baseline. Brain endothelial cells carrying APOE ε4 show higher oxidative phosphorylation, greater mitochondrial reactive oxygen species production, higher inflammatory cytokine output, and more immune cell adhesion than APOE ε3 cells [12]. In practical terms, that suggests the ε4 brain vasculature may already be closer to dysfunction before an added environmental stressor arrives.
Experimental models consistently show that small plastic particles can disrupt BBB function, and separate human and animal studies show that APOE ε4 weakens BBB integrity through its own vascular and inflammatory pathways. What remains uncertain is whether environmental microplastics and nanoplastics exploit an APOE ε4 weakened barrier more readily than they do in APOE ε3 carriers.
What remains uncertain is whether environmental microplastics and nanoplastics exploit an APOE ε4 weakened barrier more readily than they do in APOE ε3 carriers. That idea is biologically coherent, but it has not yet been directly tested in humans.
The Protein Corona and ApoE-Mediated Particle Delivery
One of the most important ideas in particle biology is that particles do not move through the body as bare material for very long. The moment they enter blood, cerebrospinal fluid, or other biological environments, they begin to pick up a coating of proteins, lipids, and other molecules. This coating is called the biomolecular corona, or more simply, the protein corona. It matters because it helps determine how the body sees the particle, where that particle travels, which cells interact with it, and how easily it crosses biological barriers.
Apolipoproteins, including ApoE, are among the proteins that can become part of that surface coating. That makes ApoE relevant not only as a brain lipid transport protein, but also as a possible guide for how certain particles move through the body.
This principle is well established in nanomedicine and increasingly relevant to environmental particle toxicology. In mouse serum experiments, polystyrene nanoparticles formed protein and lipid coronas that changed over time and varied by particle size, with apolipoproteins among the most prominent bound proteins [13].
Drug delivery research has gone a step further by deliberately using this biology. In some studies, researchers created artificial apolipoprotein coronas by adsorbing ApoE4 onto stabilized nanoparticles. That modification increased nanoparticle accumulation in mouse brains through LDL receptor mediated transcytosis [14]. The importance of that finding is not that environmental nanoplastics behave exactly the same way. It is that ApoE can materially change whether and how particles are delivered to the brain.
Kopatz and colleagues add another layer to this picture. Their work suggests that corona composition helps shape both barrier interactions and the thermodynamics of membrane entry, meaning the molecules coating a particle may influence not just recognition, but the actual ease with which it crosses into tissue [10].
ApoE binding to nanoparticles is real, and in engineered systems it can meaningfully influence central nervous system uptake. That creates a strong biological rationale for asking whether environmental nanoplastics might also be steered in an isoform dependent way. Whether ApoE4 specifically changes the brain delivery of real world environmental nanoplastics has not yet been shown. For now, this remains a mechanistic hypothesis rather than a demonstrated human pathway.
Neuroinflammation and oxidative stress
One of the most consistent themes in the microplastics literature is inflammation. Once very small particles enter tissue or are taken up by cells, they can trigger a familiar stress response: rising reactive oxygen species, impaired mitochondrial function, activation of microglia and astrocytes, and the release of inflammatory cytokines such as TNF-α, IL-1β, and IL-6. In the brain, that matters because chronic low grade neuroinflammation is not just a side effect of disease. It is a central feature of Alzheimer’s pathophysiology.
APOE ε4 is relevant here because it appears to shift that inflammatory baseline before any external exposure is added. Glial cells carrying APOE ε4 tend to be more metabolically strained, more prone to lipid droplet accumulation, more likely to generate excess reactive oxygen species, and more easily pushed into inflammasome activation, including through NLRP3 related pathways [5]. In practical terms, that suggests ε4 brains may begin closer to the threshold of inflammatory dysfunction.
Across animal studies, polystyrene nanoplastics have repeatedly been shown to activate microglial inflammatory programs, with downstream increases in IL-6, TNF-α, and NF-κB signaling. Together, these findings suggest that very small plastic particles can act as cellular stressors in the nervous system.
The most direct evidence so far for an APOE microplastics interaction comes from a 2025 study by Gaspar and colleagues. In that experiment, humanized APOE3 and APOE4 knock-in mice were exposed orally for three weeks to a mixture of 0.1 µm and 2 µm polystyrene particles. The effects differed by both genotype and sex. APOE4 mice developed behavioral and cognitive changes that were not seen in APOE3 controls, including apathy like behavior and altered risk avoidance in males, and marked recognition memory impairment in females. The study also found changes in glial markers, including GFAP and IBA1, along with shifts in liver CYP1A1 expression that suggest genotype may influence not only neuroimmune responses, but also broader xenobiotic handling [15].

A separate line of evidence points to another Alzheimer’s relevant pathway. In mice, intranasal exposure to polystyrene nanoplastics has been shown to impair glymphatic clearance of amyloid beta and phosphorylated tau by disrupting AQP4 polarization in astrocytic endfeet [16]. That matters because the glymphatic system is one of the brain’s key waste clearance routes, and reduced clearance of these proteins is already central to neurodegenerative disease models.
The strongest part of the evidence is that the APOE background appears capable of changing the brain’s response to microplastic exposure. The Gaspar study is especially important because it is, so far, the clearest peer reviewed experiment directly comparing APOE3 and APOE4 responses to microplastics in vivo. Its results do not settle the question, but they do provide proof of concept that genotype can shape behavioral and neuroimmune responses to exposure.
Whether similar effects occur under chronic, mixed polymer, environmentally realistic exposure conditions remains unknown. The sex specific findings also raise important questions about hormonal influences that are not yet resolved.
Protein aggregation and proteostasis
Another important overlap between Alzheimer’s biology and microplastic research involves proteostasis, the set of cellular systems that keep proteins properly folded, trafficked, and cleared. In Alzheimer’s disease, that balance breaks down. Amyloid beta peptides begin to misfold and aggregate, tau becomes pathologically altered, and the brain’s clearance systems struggle to keep pace.
APOE ε4 already sits inside this problem. Compared with ApoE3 or ApoE2, ApoE4 is less effective at helping clear amyloid beta from the brain’s interstitial fluid. Over time, that may contribute to greater amyloid buildup and a more permissive environment for protein aggregation.
A separate line of evidence suggests that very small plastic particles may add pressure to this same system. In laboratory models, nanoplastics have been shown to interact directly with aggregation prone proteins and in some cases appear to speed the transition from soluble proteins to misfolded fibrils. One proposed explanation is that the particle surface acts as a kind of scaffold or nucleation site, increasing the likelihood that vulnerable proteins will misfold and cluster together.
That possibility has now been shown across more than one protein system. Anionic nanoplastics have been found to promote alpha synuclein fibrillation by binding to the protein’s NAC region, shortening the lag time before aggregation begins and slowing lysosomal degradation of the resulting material [17]. Low concentrations of polystyrene nanoplastics have also been shown to accelerate amyloid beta 40 and 42 nucleation and increase neurotoxicity in vitro [18]. These findings do not prove that the same process is happening in the human brain, but they do suggest that nanoplastics can influence the physical chemistry of protein aggregation under experimental conditions.
Animal work adds a second layer. In Alzheimer’s disease mouse models, polystyrene microplastics have been shown to worsen cognitive decline while activating microglial pyroptosis and inflammatory signaling through the NLRP3, Caspase 1, and NF-κB pathways [19]. That matters because impaired protein clearance and inflammation often reinforce one another in neurodegeneration.
There is also evidence that microplastics and nanoplastics may interfere with the cell’s internal waste handling systems more broadly. When these particles accumulate in lysosomes, the cell cannot readily degrade them. That may disrupt autophagic flux, one of the key pathways cells use to clear damaged components and protein aggregates. In that sense, the issue may not be only whether nanoplastics help initiate aggregation, but whether they also make it harder for the cell to clean up once aggregation begins.
Nanoplastics can accelerate aggregation related processes in vitro, and animal models show signs of both neuroinflammatory stress and impaired protein handling after exposure. What remains uncertain is whether environmental microplastics and nanoplastics actually seed amyloid beta aggregation in the human brain under real world conditions. At this stage, the mechanism is plausible, but unproven.
The gut microbiome brain axis
The gut microbiome brain axis refers to the constant two way communication between the gastrointestinal tract and the central nervous system. That communication is shaped in part by gut bacteria, which help influence immune tone, inflammatory signaling, neurotransmitter related pathways, and even the integrity of the blood brain barrier. Some of these effects are mediated through short chain fatty acids, or SCFAs, along with vagal signaling and broader immune crosstalk. When the microbiome shifts out of balance, that disruption, often called dysbiosis, has been increasingly linked to neurodegenerative disease, including both Alzheimer’s and Parkinson’s.
APOE ε4 may matter here earlier than many people realize. Even in young and otherwise healthy adults, APOE ε4 carriers appear to show measurable differences in gut microbiome composition. In a 2025 study by Bressa and colleagues, healthy APOE ε4 carriers showed a roughly fivefold reduction in Megamonas, a genus associated with energy metabolism and SCFA production [20]. Earlier work by Tran and colleagues also found that APOE genotype shapes gut microbial structure in both humans and mice, with APOE ε4 linked to reduced Prevotellaceae and altered SCFA profiles [21]. These findings suggest that APOE ε4 may set a different microbial baseline long before overt neurological disease appears.
Microplastics enter this picture from another direction. Across animal models, ingested microplastics have been shown to disrupt the intestinal barrier, alter microbial composition, and reduce populations of beneficial SCFA producing organisms such as Lactobacillus and Ruminococcus. In other words, they can weaken the gut lining while also shifting the microbial communities that help maintain it. In Parkinson’s disease models, polystyrene microplastics have been shown to worsen gut barrier damage, reduce beneficial bacteria, and contribute to dopaminergic neuron degeneration through gut brain axis related pathways [22].
Human data support that APOE ε4 is associated with a distinct baseline microbiome profile, and animal data support that microplastics can independently drive dysbiosis and barrier disruption. What remains uncertain is whether those two vulnerabilities interact in a way that meaningfully amplifies neurological risk. For now, that remains a synthesis of adjacent evidence rather than a directly proven mechanism.
Human evidence
The human evidence on microplastics, nanoplastics, and neurological risk is still early. It is limited, observational, and needs to be described with real care.

What human studies can tell us at this stage is that these particles are showing up in places that matter. Researchers have detected microplastics and nanoplastics in human blood, olfactory bulbs, carotid artery plaques, and post mortem brain tissue using methods such as pyrolysis GC MS, micro FTIR, and Raman spectroscopy [1,23]. That does not, on its own, prove harm. But it does confirm internal exposure and tissue presence, including in sites relevant to vascular and neurological health.
In brain tissue studies, the key unresolved issue is interpretation rather than detection. Higher microplastic concentrations have been reported in samples from individuals with documented dementia than in controls, but that finding does not tell us which direction the relationship runs. Microplastics could be contributing to disease processes, or dementia related barrier dysfunction could allow greater accumulation over time. At present, the human data cannot separate those possibilities.
The strongest human outcome association in the broader literature comes from cardiovascular disease, not neurodegeneration. In one study, the presence of microplastics and nanoplastics in carotid artery plaques was associated with a roughly 4.5 fold higher risk of stroke, myocardial infarction, or death during follow up [24]. That is an important signal. But it is still observational, which means it can identify association, not prove cause.
What human evidence does not yet show is just as important. There is currently no longitudinal study linking internal microplastic burden to the later development of Alzheimer’s disease or other neurodegenerative outcomes. There is no epidemiologic dataset stratifying microplastic related health outcomes by APOE genotype. And there is no human cohort in which causality has been established.
That leaves the field in an important but unresolved position. The human data are enough to justify concern and much more research. They are not yet enough to claim that microplastics cause Alzheimer’s disease, or that APOE ε4 carriers are already known to face higher neurological harm from exposure. For now, the human literature tells us that exposure is real, accumulation is plausible, and clinical relevance remains an open question.
Methodological limitations
The central question is scientifically important, but the methods are still catching up to the complexity of the real world.
Exposure realism. The most important direct study of an APOE microplastics interaction, the 2025 paper by Gaspar and colleagues, used pristine, uniform, fluorescently labeled polystyrene spheres at concentrations above current estimates of typical human internal exposure [15]. That kind of design is useful for testing whether an effect is possible under controlled conditions. But it is not the same as everyday human exposure, which is likely to involve a lifelong mix of low dose ingestion and inhalation from many particle types at once. The gap between laboratory exposure and real world exposure remains large.
Pristine versus weathered plastics. The plastics people encounter outside the lab are not clean, untouched spheres. Environmental particles are weathered by sunlight, friction, oxidation, and mechanical wear. They may also carry adsorbed metals, plastic additives such as phthalates and bisphenols, and microbial films. That matters because some observed toxicity may come not only from the polymer itself, but also from the chemical cargo attached to it. This is sometimes described as a Trojan horse problem. In many studies, it is still difficult to separate the effects of the particle from the effects of what the particle is carrying. And because weathered plastics behave differently from pristine ones, that distinction matters more than it is often given credit for [25].

Measurement constraints. The smallest particles may be the most biologically important, but they are also the hardest to measure. Reliable detection of true nanoplastics, especially below 100 nm, in complex human tissues remains technically difficult. Methods such as py-GC-MS can estimate polymer mass, but they do not by themselves provide clear information about particle number, shape, or size distribution without additional imaging methods. Contamination control is also a major challenge. Even in the recent autopsy brain study, the researchers analyzed very small tissue subsamples, around 1 to 2 mg, and sampled only a single site per organ for each individual [1]. That does not invalidate the findings, but it does place important limits on how confidently they can be generalized.
Particle type specificity. Most experimental neurotoxicity work relies on polystyrene beads because they are commercially standardized, easy to label, and easier to track in biological systems. But human tissue studies often report a different pattern, with polyethylene appearing especially common. That raises an obvious question: do findings from polystyrene translate to polyethylene, polypropylene, or mixed polymer exposure? At this stage, that cannot be assumed.
Model translation. Even the best animal models remain approximations. The Gaspar study used mice that were 3 to 6 months old and exposed them for only 3 weeks as a stand-in for what, in humans, would likely be decades of cumulative exposure. Humanized knock-in mice are valuable tools, but they do not fully reproduce human APOE biology, human aging, hormonal context, or the full complexity of lifelong exposure. And while mouse behavioral tests can be informative, they are still imperfect stand-ins for human cognition and neurodegenerative disease.
Single study for the critical interaction. Perhaps the most important limitation is that the direct APOE microplastics interaction evidence still rests on a single peer reviewed study. That study is important, and genuinely informative. But one study is not enough to anchor strong mechanistic conclusions. Replication is the minimum standard needed before this interaction can be treated as established rather than suggestive.
Taken together, these limitations define the level of confidence the current evidence can bear. The field is still early, and the strongest claims remain ahead of the data.
Synthesis
Taken together, the literature points to a coherent pattern, even if it does not yet establish causation.
APOE ε4 is associated with a well described pattern of vulnerability across several systems already central to neurodegeneration. Microplastics and nanoplastics, especially at smaller sizes, appear to affect many of those same systems in laboratory and animal studies.
The most reasonable interpretation of the evidence so far is that APOE ε4 may create a biological terrain that is less resilient to microplastic and nanoplastic related stress than APOE ε3. The 2025 Gaspar study offers an early proof of concept for that idea in mice. The protein corona literature supports it at the level of particle transport and brain delivery. The gut microbiome literature supports it at the systems level. None of that proves the same pattern is already occurring in humans. But it does make the hypothesis more than arbitrary.
That distinction matters. Mechanistically plausible and supported by early animal data is not the same as demonstrated in humans. The gap between those two levels of evidence is still wide.
What the current evidence does not justify is dismissal. When multiple independent lines of research begin converging on the same biologically vulnerable population, that is usually not the moment to wave the question away. It is the moment to investigate it with more precision, better tools, and greater urgency.
References
- 1.↑ Nihart, A. J. et al. Bioaccumulation of microplastics in decedent human brains. Nat. Med. 31, 1114–1119 (2025). AtlasPubMed
- 2.↑ Corder, E. H. et al. Gene Dose of Apolipoprotein E Type 4 Allele and the Risk of Alzheimer’s Disease in Late Onset Families. Science 261, 921–923 (1993). PubMed
- 3.↑ Mahley, R. W., Weisgraber, K. H. & Huang, Y. Apolipoprotein E: structure determines function, from atherosclerosis to Alzheimer’s disease to AIDS. J. Lipid Res. 50, S183–S188 (2009). PubMed
- 4.↑ Bell, R. D. et al. Apolipoprotein E controls cerebrovascular integrity via cyclophilin A. Nature 485, 512–516 (2012). PubMed
- 5.↑ TCW, J. et al. Cholesterol and matrisome pathways dysregulated in astrocytes and microglia. Cell 185, 2213-2233.e25 (2022). PubMed
- 6.↑ Zhao, N., Liu, C.-C., Qiao, W. & Bu, G. Apolipoprotein E, Receptors, and Modulation of Alzheimer’s Disease. Biol. Psychiatry 83, 347–357 (2018). PubMed
- 7.↑ Amato-Lourenço, L. F. et al. Microplastics in the Olfactory Bulb of the Human Brain. JAMA Netw. Open 7, e2440018 (2024). AtlasPubMed
- 8.↑ Shan, S., Zhang, Y., Zhao, H., Zeng, T. & Zhao, X. Polystyrene nanoplastics penetrate across the blood-brain barrier and induce activation of microglia in the brain of mice. Chemosphere 298, 134261 (2022). AtlasPubMed
- 9.↑ Jin, H. et al. Evaluation of Neurotoxicity in BALB/c Mice following Chronic Exposure to Polystyrene Microplastics. Environ. Heal. Perspect. 130, 107002 (2022). AtlasPubMed
- 10.↑ Kopatz, V. et al. Micro- and Nanoplastics Breach the Blood–Brain Barrier (BBB): Biomolecular Corona’s Role Revealed. Nanomaterials 13, 1404 (2023). PubMed
- 11.↑ Montagne, A. et al. APOE4 leads to blood–brain barrier dysfunction predicting cognitive decline. Nature 581, 71–76 (2020). PubMed
- 12.↑ Marottoli, F. M. et al. Autocrine Effects of Brain Endothelial Cell-Produced Human Apolipoprotein E on Metabolism and Inflammation in vitro. Front. Cell Dev. Biol. 9, 668296 (2021). PubMed
- 13.↑ Lima, T., Bernfur, K., Vilanova, M. & Cedervall, T. Understanding the Lipid and Protein Corona Formation on Different Sized Polymeric Nanoparticles. Sci. Rep. 10, 1129 (2020). PubMed
- 14.↑ Topal, G. R. et al. ApoE-Targeting Increases the Transfer of Solid Lipid Nanoparticles with Donepezil Cargo across a Culture Model of the Blood–Brain Barrier. Pharmaceutics 13, 38 (2020). PubMed
- 15.↑ Gaspar, L., Bartman, S., Tobias-Wallingford, H., Coppotelli, G. & Ross, J. M. Short-term exposure to polystyrene microplastics alters cognition, immune, and metabolic markers in an apolipoprotein E (APOE) genotype and sex-dependent manner. Environ. Res. Commun. 7, 10.1088/2515-7620/adf8ae (2025). AtlasPubMed
- 16.↑ Sun, M. et al. Polystyrene nanoplastics induced learning and memory impairments in mice by damaging the glymphatic system. Ecotoxicol. Environ. Saf. 284, 116874 (2024). AtlasPubMed
- 17.↑ Liu, Z. et al. Anionic nanoplastic contaminants promote Parkinson’s disease–associated α-synuclein aggregation. Sci. Adv. 9, eadi8716 (2023). AtlasPubMed
- 18.↑ Gou, X. et al. Impact of nanoplastics on Alzheimer ’s disease: Enhanced amyloid-β peptide aggregation and augmented neurotoxicity. J. Hazard. Mater. 465, 133518 (2024). AtlasPubMed
- 19.↑ Wang, G., Lin, Y. & Shen, H. Exposure to Polystyrene Microplastics Promotes the Progression of Cognitive Impairment in Alzheimer’s Disease: Association with Induction of Microglial Pyroptosis. Mol. Neurobiol. 61, 900–907 (2024). AtlasPubMed
- 20.↑ Bressa, C. et al. Exploring the relationship between APOEε4 allele and gut microbiota composition and function in healthy adults. AMB Express 15, 77 (2025). PubMed
- 21.↑ Tran, T. T. T. et al. APOE genotype influences the gut microbiome structure and function in humans and mice: relevance for Alzheimer’s disease pathophysiology. FASEB J. 33, 8221–8231 (2019). PubMed
- 22.↑ Bai, H. et al. PD-like pathogenesis induced by intestinal exposure to microplastics: An in vivo study of animal models to a public health survey. J. Hazard. Mater. 486, 136974 (2025). AtlasPubMed
- 23.↑ Nardella, F. et al. Advancing pyrolysis-gas chromatography-mass spectrometry for the accurate quantification of micro- and nanoplastics in human blood. Microplastics Nanoplastics 5, 48 (2025). PubMed
- 24.↑ Marfella, R. et al. Microplastics and Nanoplastics in Atheromas and Cardiovascular Events. N. Engl. J. Med. 390, 900–910 (2024). AtlasPubMed
- 25.↑ Alimi, O. S. et al. Weathering pathways and protocols for environmentally relevant microplastics and nanoplastics: What are we missing? J. Hazard. Mater. 423, 126955 (2022). AtlasPubMed
Sign in to start a discussion.