When “debunking” becomes dismissal
Structural asymmetries in microplastic risk assessment
Research on micro- and nanoplastics is entering a period of methodological refinement. This is a normal stage in the development of any emerging scientific field. Early measurements are questioned. Methods improve. The picture becomes clearer over time.
Several early exposure estimates have been revised after methodological concerns were identified. Some studies relied on particle to mass conversions considered too coarse. Others used inconsistent particle size assumptions or lacked sufficient contamination controls. These are common growing pains in environmental measurement science. Correcting them strengthens the field. It does not weaken it.
A different interpretation has begun to appear alongside these corrections. Some commentators now suggest that revised exposure estimates imply that microplastic exposure is unlikely to be biologically meaningful [1]. That conclusion extends beyond what the evidence can support.
Scientific uncertainty is not directional. When evidence is incomplete, it cannot confidently confirm harm. But neither can it confidently confirm safety.
This paper takes a more pragmatic view. Microplastic risk cannot be evaluated through ingestion and accumulation mass alone. Detection limits do not establish absence. And the standard of proof often expected in environmental health science is rarely achievable when exposures are chronic, diffuse, and ethically difficult to definitively test in humans.
At the same time, evidence is accumulating across several independent research approaches. Environmental monitoring studies detect plastic particles in human samples [2-7]. Controlled laboratory experiments observe biological responses in cell and animal models. Mechanistic studies describe how particles interact with cells, tissues, and biochemical pathways.

Taken together, these lines of work point in a consistent direction. Micro- and nanoplastics are not biologically inert. They interact with living systems.
Long term human trials do not yet exist. That absence reflects the limits of environmental health science, not evidence of safety.
The question, therefore, is not whether the science is finished. It is whether the signals that already exist deserve careful attention. The remaining uncertainty is not about the existence of risk, but about its magnitude.
Scientific self-correction is not scientific failure
Skeptics of microplastic exposure research have raised valid methodological concerns [8-14]. Several early ingestion estimates relied on simplified particle to mass assumptions. Some studies underestimated contamination risks during sampling or laboratory processing. In a few cases, analytical techniques struggled to distinguish plastics from biological material.
This is the normal course of scientific progress. Many of these limitations were identified and openly discussed by the researchers themselves. Acknowledging and addressing them has helped refine methods, strengthen analytical standards, and ultimately improve the quality of the field [16].
Laboratories are working to employ stricter procedural blanks, cleaner sampling environments, and more careful spectroscopic identification methods. Updated exposure models incorporating these improvements suggest that annual dietary ingestion mass may fall in the microgram to milligram range for typical individuals.
Those revisions are useful. They provide a more realistic estimate of total exposure mass.
But ingestion mass is only one part of the story.
Environmental toxicology has long recognized that particulate materials behave differently from dissolved chemicals [16-22]. Their biological effects often depend on particle number, size, surface area, and chemistry as much as on total mass. For this reason, the implications of plastic particle exposure cannot be evaluated through ingestion mass alone. The picture is further complicated by stochastic absorption and the potential for long term accumulation, which differ fundamentally from the more predictable dose response patterns typically associated with small molecule toxins.
More importantly, ingestion estimates represent only one of several ways the problem is being studied. Microplastics are now examined simultaneously through environmental detection studies, experimental exposure models, and mechanistic biological research. No single approach provides a complete answer. Together they form a broader evidence landscape.
Why mass alone cannot describe particulate risk
Mass is an intuitive metric. It works well for many chemical exposures.
For particles, however, it can be misleading.
Ultrafine air pollution, asbestos fibers, and engineered nanoparticles all demonstrate the same principle. Biological responses often correlate more strongly with particle number, surface area, and geometry than with total mass.
Microplastics follow similar logic.
A single particle measuring one hundred micrometers contains roughly the same mass as billions of particles measuring one hundred nanometers. Yet those two exposures present very different biological scenarios.

As particles become smaller, surface area per unit mass increases sharply. Their ability to absorb environmental contaminants rises. Cellular uptake becomes more likely. Interactions with immune cells and epithelial barriers become possible.
Nanoplastics appear capable of crossing biological barriers through mechanisms such as endocytosis and paracellular transport. Some may interact directly with immune tissues in the gut.
Detection technologies, however, remain better suited to larger particles. Many analytical techniques struggle to measure plastics at the nanoscale.
This creates an important blind spot. The particles most capable of interacting with biological systems may also be the hardest to measure directly.
For this reason, ingestion mass alone cannot define biological risk. Other lines of evidence become essential.
Experimental models reveal consistent biological interaction
Laboratory studies provide one of those lines.
Across a growing body of work, researchers have examined how microplastics interact with living organisms. The findings are not uniform in magnitude or interpretation, but certain patterns appear repeatedly.
In gastrointestinal models [23-101], plastic particles have been shown to alter intestinal barrier function and microbial communities. Mouse studies have reported disruption of tight junction proteins and shifts in gut microbiota composition following exposure to polystyrene particles.
These changes are frequently accompanied by inflammatory signaling. Reductions in beneficial bacterial groups such as Lactobacillaceae and Bifidobacteriaceae have been observed alongside increases in pro-inflammatory taxa.
Other studies describe systemic effects [102-113]. Animal models have documented oxidative stress, lipid metabolism disruption, and inflammatory responses in liver and pancreatic tissue after chronic ingestion of plastic particles.
Developmental models add another layer [114-130]. Zebrafish studies examining multigenerational exposure have reported metabolic changes and reproductive effects that persist across generations. And mice models further strengthen those results.
The details vary across species and experimental designs. What stands out is the recurrence of biological interaction.
Similar responses have now been reported in insects, worms, fish, rodents, and other vertebrate systems. The organisms differ. The exposure pathways differ. Yet the signal appears again and again: plastic particles are capable of interacting with living tissues.
Plastic surfaces also behave chemically. Laboratory work shows that micro- and nanoplastics are made with and can adsorb from their environment other concerning pollutants [131-144], like heavy metals, flame retardants, persistent organic pollutants, antibiotic resistant bacteria. Once ingested, those compounds may become more bioavailable.
In this way, plastic particles function not only as physical materials but also as carriers.
Early signals in human studies
Human evidence remains early, but it is growing.
Microplastic particles have now been identified in several human tissues and biological fluids. Studies have reported plastic polymers in blood samples, while other researchers have detected plastic fragments in placental tissue and amniotic fluid collected from otherwise healthy pregnancies, in uterine fibroids, and effects on semen count and quality and [145-151].
More recently, investigators examining atherosclerotic plaques obtained during vascular surgery identified micro and nanoplastic particles in a subset of samples [152]. During follow up, individuals whose plaques contained detectable plastic particles experienced higher rates of cardiovascular events than those without detectable particles.
These findings do not establish causality. They do establish something important. Plastic particles are capable of entering the human body and reaching internal tissues.
Additional evidence comes from in vitro research using human cells [153-159]. In these experimental systems, many of the biological responses observed in animal models—such as oxidative stress, inflammatory signaling, and cellular disruption—are also observed in human cell lines.
Taken together, these findings form a consistent pattern. Detection studies confirm exposure. Experimental models observe biological responses. Mechanistic studies describe pathways through which those responses may occur. As these lines of evidence accumulate, the overall picture becomes increasingly coherent.
Detection limits and the illusion of absence
Detecting plastics within biological samples remains technically challenging, particularly as particle size decreases.
Human tissues contain lipids, proteins, and other organic material that can interfere with spectroscopic measurements. Polymer identification therefore requires careful sample preparation, strict contamination controls, and analytical methods capable of distinguishing plastics from complex biological matrices. Nanoplastic particles present an additional challenge, as many fall below the detection limits of current instrumentation.
For these reasons, non detection should not be interpreted as proof of absence.
Environmental health science has encountered this dynamic many times before [160-168]. Numerous contaminants were initially underestimated until improvements in analytical chemistry revealed exposure pathways that earlier methods were simply unable to measure. Microplastics may be following a similar trajectory.
The recent discussion surrounding pyrolysis GC–MS provides a useful example. Some critics have pointed out that certain biological lipids can produce fragments that resemble polymer signatures, raising the possibility of overestimation when samples are not carefully prepared. At the same time, methodological limitations can also lead to underestimation, particularly when smaller particles or complex biological matrices obscure detection.
In practice, measurement uncertainty places the true value somewhere between these bounds. This is where the distinction between precision and accuracy becomes important. Analytical methods may produce values that are reproducible, but still incomplete representations of the full exposure landscape.
Estimating exposure with confidence is difficult. Demonstrating that exposure is zero is harder still.
The structural lag in environmental health science
The debate surrounding microplastics reflects a broader pattern seen repeatedly in environmental health science.
Persistent materials are frequently introduced into the environment under an initial assumption of safety. Evidence of harm often accumulates gradually, and by the time clear epidemiological signals emerge in human populations, exposure may already be widespread.
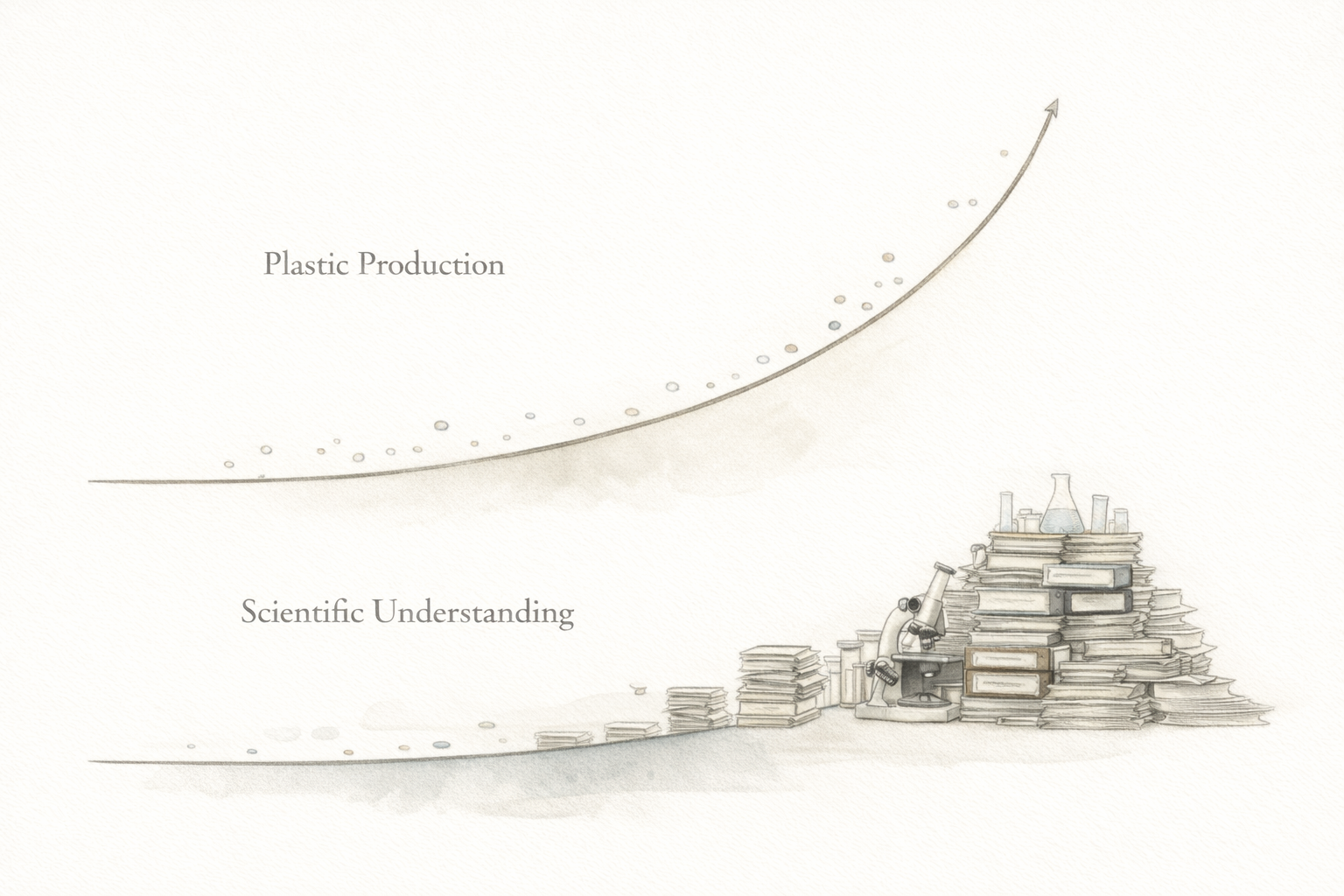
This dynamic is not limited to environmental contaminants. Comparable patterns have occurred in other areas of public health, including the delayed recognition of the cardiovascular risks associated with industrial trans fats [169–172].
Scientists face fundamental barriers when investigating these questions. Controlled exposure experiments cannot ethically be performed in humans. High dose, short term studies are often criticized as unrealistic, while mechanistic research is sometimes dismissed as hypothetical. Observational studies, meanwhile, require long time horizons and very large populations to detect meaningful signals. At the same time, widespread background contamination increasingly complicates the identification of truly unexposed populations. When pollution becomes pervasive, meaningful negative control groups may effectively disappear.
Environmental health science has encountered this dynamic many times before. The risks associated with lead, tobacco smoke, airborne particulate matter, certain pesticides, and industrial trans fats only became clear after years or decades of accumulating evidence.
The result is an inherent asymmetry.
Industrial deployment moves quickly and is often granted the benefit of the doubt. Scientific certainty develops more slowly and is subject to far greater scrutiny, particularly when potential findings carry economic implications.
It can often feel like an evershifting goal post.

This pattern is familiar in environmental history. New materials are widely adopted under a presumption of safety, while evidence of potential harm must meet a much higher threshold before it meaningfully alters industrial practice.
Historically, early signals of risk tend to appear first in mechanistic research and experimental models. Human epidemiology often follows years or decades later.
Microplastic research may now be entering that earlier stage.
Why precaution becomes rational
Several points are already clear.
Plastic production continues to increase globally. Environmental contamination is widespread. Human exposure is occurring.
Experimental studies consistently show that plastic particles interact with biological systems. Inflammatory responses, microbiome shifts, and metabolic changes have been observed across multiple models.
At the same time, important questions remain unanswered. Long term dose response relationships are not yet known. The distribution of nanoplastics within human tissues is poorly understood.
Uncertainty remains.
But uncertainty is not the same as safety.
When multiple independent lines of research point toward biological interaction, careful investigation becomes the rational response.
Public expectations and environmental health
Public concern about environmental exposures reflects this history.
Recent surveys indicate that a large majority of people worry about harmful chemicals in food, water, and consumer products [173-175]. Many believe regulatory systems should do more to evaluate emerging contaminants.
This concern is not simply fear. It reflects decades of experience with environmental hazards that were recognized only after long delays.
Observation over dismissal
Correcting exaggerated claims improves scientific clarity.
But correction should not be mistaken for dismissal.
Mass based exposure estimates provide useful information, yet they capture only part of the biology of particulate materials. Plastic particles interact with cells, tissues, microbes, and chemical environments in ways that mass alone cannot describe.
Across environmental monitoring, experimental models, and emerging human studies, a consistent signal is appearing. Microplastics are biologically interactive materials. Their long term consequences remain uncertain.
The appropriate scientific response is neither alarm nor denial. It is careful observation.
Where exposure can be reduced, reduction is reasonable. Where mitigation strategies exist, they deserve investigation.
Environmental health science rarely provides certainty quickly. Waiting for absolute proof has often meant waiting too long.
Precaution, in this context, is not panic. It is simply the recognition that people are allowed to respond thoughtfully to the information available to them. Some may choose to reduce personal exposure. Others may continue largely as they always have while watching the science develop. Some will advocate for systemic change. Others will focus on practical steps within their own lives.
None of these responses are unreasonable.
The world rarely presents issues in simple black and white terms. Environmental health questions often unfold in shades of gray. What matters is that people can examine the evidence, think critically about what it means, and engage in open, informed conversation.
Science advances this way. By continuing to study. Continuing to question. Continuing to learn. At its core, science reflects a deeply human process: observing the world, testing ideas, updating our understanding, and moving forward with better information.
Precaution, when grounded in evidence, is not fear. It is patience informed by data. It is the freedom to act in ways that feel reasonable with the knowledge available today, while remaining open to what tomorrow’s research may reveal.
References
- 1.↑ Nor, N. H. M., Kooi, M., Diepens, N. J. & Koelmans, A. A. Lifetime Accumulation of Microplastic in Children and Adults. Environ. Sci. Technol. 55, 5084–5096 (2021). PubMed
- 2.↑ Hu, C. J. et al. Microplastic presence in dog and human testis and its potential association with sperm count and weights of testis and epididymis. Toxicol. Sci. 200, 235–240 (2024). PubMed
- 3. Ragusa, A. et al. Plasticenta: First evidence of microplastics in human placenta. Environ. Int. 146, 106274 (2021). PubMed
- 4. Leslie, H. A. et al. Discovery and quantification of plastic particle pollution in human blood. Environ. Int. 163, 107199 (2022). PubMed
- 5. Ragusa, A. et al. Raman Microspectroscopy Detection and Characterisation of Microplastics in Human Breastmilk. Polymers 14, 2700 (2022). PubMed
- 7.↑ Marfella, R. et al. Microplastics and Nanoplastics in Atheromas and Cardiovascular Events. N. Engl. J. Med. 390, 900–910 (2024). PubMed
- 8.↑ Monikh, F. A. et al. Challenges in studying microplastics in human brain. Nat. Med. 31, 4034–4035 (2025). https://pubmed.ncbi.nlm.nih.gov/41233597/; Campen, M. J., West, A. B., Garcia, M., Gullapalli, R. & Hayek, E. E. Reply to: Challenges in studying microplastics in human brain. Nat. Med. 31, 4036–4037 (2025). PubMed
- 9. Kalkman, D. N., Renkens, M. P. L. & Grundeken, M. J. Microplastics and Nanoplastics in Atheromas. N. Engl. J. Med. 390, 1726–1728 (2024). PubMed
- 10. Uppu, R. M., Peijnenburg, W. & Hays, S. M. Comment on: “Microplastic presence in dog and human testis and its potential association with sperm count and weights of testis and epididymis.” Toxicol. Sci. 206, 456–457 (2024). https://pubmed.ncbi.nlm.nih.gov/39475449/; Hu, C. J. et al. Response to comment on: “Microplastic presence in dog and human testis and its potential association with sperm count and weights of testis and epididymis.” Toxicol. Sci. 206, 458–459 (2024). PubMed
- 11. Brits, M. et al. Response on the commentary by B. Wilhelmus, M. Gahleitner, and M. A. Pemberton, on the manuscript by M. Brits, M.J.M. van Velzen, F.Ö. Sefiloglu, L. Scibetta, Q. Groenewoud, J.J. Garcia-Vallejo, A.D. Vethaak, S.H. Brandsma, M.H. Lamoree. Quantitation of micro and nanoplastics in human blood by pyrolysis-gas chromatography–mass spectrometry: a follow-up study. Microplastics and Nanoplastics (2024) 4:12. Microplastics Nanoplastics 4, 29 (2024).
- 12. Materić, D. Nanoplastics measurements must have appropriate blanks. Proc. Natl. Acad. Sci. 121, e2411099121 (2024). https://pubmed.ncbi.nlm.nih.gov/39546557/; Qian, N., Stapleton, P., Yan, B. & Min, W. Reply to Materić: Appropriate blanks should avoid major contamination sources in the lab. Proc. Natl. Acad. Sci. 121, e2415874121 (2024). PubMed
- 13. Janzik, R., Sieg, H., Braeuning, A. & Böl, G.-F. Microplastics: State of the evidence on health effects and public perception. Dtsch. Ärzteblatt Int. 122, 546–551 (2025). PubMed
- 14.↑ Kuhlman, R. L. Letter to the editor, discovery and quantification of plastic particle pollution in human blood. Environ. Int. 167, 107400 (2022). PubMed
- 15. Rauert, C. et al. Assessing the Efficacy of Pyrolysis–Gas Chromatography–Mass Spectrometry for Nanoplastic and Microplastic Analysis in Human Blood. Environ. Sci. Technol. 59, 1984–1994 (2025). PubMed
- 16.↑ Oberdörster, G., Oberdörster, E. & Oberdörster, J. Nanotoxicology: An Emerging Discipline Evolving from Studies of Ultrafine Particles. Environ. Heal. Perspect. 113, 823–839 (2005). PubMed
- 17. Donaldson, K. & Stone, V. Current hypotheses on the mechanisms of toxicity of ultrafine particles. Ann. dell’Ist. Super. di sanita 39, 405–10 (2003). PubMed
- 18. Riediker, M. et al. Particle toxicology and health - where are we? Part. Fibre Toxicol. 16, 19 (2019). PubMed
- 19. Borm, P. J. et al. The potential risks of nanomaterials: a review carried out for ECETOC. Part. Fibre Toxicol. 3, 11 (2006). PubMed
- 20. Donaldson, K., Poland, C. A. & Schins, R. P. F. Possible genotoxic mechanisms of nanoparticles: Criteria for improved test strategies. Nanotoxicology 4, 414–420 (2010). PubMed
- 21. Donaldson, K. et al. Pulmonary toxicity of carbon nanotubes and asbestos — Similarities and differences. Adv. Drug Deliv. Rev. 65, 2078–2086 (2013). PubMed
- 22.↑ Maynard, A. D., Warheit, D. B. & Philbert, M. A. The New Toxicology of Sophisticated Materials: Nanotoxicology and Beyond. Toxicol. Sci. 120, S109–S129 (2011). PubMed
- 23.↑ Chen, X. et al. Polyvinyl chloride microplastics induced gut barrier dysfunction, microbiota dysbiosis and metabolism disorder in adult mice. Ecotoxicol. Environ. Saf. 241, 113809 (2022). PubMed
- 24. Chen, W. et al. Cyanidin-3-O-glucoside impacts fecal discharge of polystyrene microplastics in mice: Potential role of microbiota-derived metabolites. Toxicol. Appl. Pharmacol. 453, 116212 (2022). PubMed
- 25. Chen, Y. et al. Polystyrene microplastics aggravate radiation-induced intestinal injury in mice. Ecotoxicol. Environ. Saf. 283, 116834 (2024). PubMed
- 26. Chen, J. et al. Multiomics Reveals Nonphagocytosable Microplastics Induce Colon Inflammatory Injury via Bile Acid-Gut Microbiota Interactions and Barrier Dysfunction. ACS Appl. Mater. Interfaces 17, 44138–44159 (2025). PubMed
- 27. Chen, D. et al. Microbial diversity and metabolomics analysis of colon contents exposed to cadmium and polystyrene microplastics. Ecotoxicol. Environ. Saf. 290, 117585 (2025). PubMed
- 28. Chi, J. et al. Metabolic Reprogramming in Gut Microbiota Exposed to Polystyrene Microplastics. Biomedicines 13, 446 (2025). PubMed
- 29. Choi, Y. S. et al. Dysbiosis of gut microbiota in C57BL/6-Lepem1hwl/Korl mice during microplastics-caused hepatic metabolism disruption. PLOS One 20, e0336627 (2025). PubMed
- 30. Deng, Y. et al. Microplastics release phthalate esters and cause aggravated adverse effects in the mouse gut. Environ. Int. 143, 105916 (2020). PubMed
- 31. Gan, H.-J. et al. Simulated Microplastic Release from Cutting Boards and Evaluation of Intestinal Inflammation and Gut Microbiota in Mice. Environ. Heal. Perspect. 133, 047004 (2025). PubMed
- 32. Gao, B. et al. Size-dependent effects of polystyrene microplastics on gut metagenome and antibiotic resistance in C57BL/6 mice. Ecotoxicol. Environ. Saf. 254, 114737 (2023). PubMed
- 33. Gao, B. et al. Mixture Effects of Polystyrene Microplastics on the Gut Microbiota in C57BL/6 Mice. ACS Omega 10, 7597–7608 (2025). PubMed
- 34. Gao, B. et al. Association between microplastics and the functionalities of human gut microbiome. Ecotoxicol. Environ. Saf. 290, 117497 (2025). PubMed
- 35. Hasegawa, Y. et al. Oral exposure to high concentrations of polystyrene microplastics alters the intestinal environment and metabolic outcomes in mice. Front. Immunol. 15, 1407936 (2024). PubMed
- 36. Hsu, W.-H. et al. Polystyrene nanoplastics disrupt the intestinal microenvironment by altering bacteria-host interactions through extracellular vesicle-delivered microRNAs. Nat. Commun. 16, 5026 (2025). PubMed
- 37. Huang, W. et al. Influence of the co-exposure of microplastics and tetrabromobisphenol A on human gut: Simulation in vitro with human cell Caco-2 and gut microbiota. Sci. Total Environ. 778, 146264 (2021). PubMed
- 38. Huang, H.-J. et al. Polystyrene Microplastics Can Aggravate the Damage of the Intestinal Microenvironment Caused by Okadaic Acid: A Prevalent Algal Toxin. Mar. Drugs 23, 129 (2025). PubMed
- 39. Hwangbo, H. et al. Polystyrene Accelerates Aging Related-Gut Microbiome Dysbiosis and -Metabolites in Old-Aged Mouse. J. Microbiol. Biotechnol. 35, 1–11 (2025). PubMed
- 40. Jeong, B. et al. Maternal nanoplastic ingestion induces an increase in offspring body weight through altered lipid species and microbiota. Environ. Int. 185, 108522 (2024). PubMed
- 41. Jiménez-Arroyo, C. et al. Simulated gastrointestinal digestion of polylactic acid (PLA) biodegradable microplastics and their interaction with the gut microbiota. Sci. Total Environ. 902, 166003 (2023). PubMed
- 42. Jin, Y., Lu, L., Tu, W., Luo, T. & Fu, Z. Impacts of polystyrene microplastic on the gut barrier, microbiota and metabolism of mice. Sci. Total Environ. 649, 308–317 (2019). PubMed
- 43. Kaluç, N. et al. Gut-lung microbiota dynamics in mice exposed to Nanoplastics. NanoImpact 36, 100531 (2024). PubMed
- 44. Ke, D. et al. Occurrence of microplastics and disturbance of gut microbiota: a pilot study of preschool children in Xiamen, China. eBioMedicine 97, 104828 (2023). PubMed
- 45. Kim, K. J. et al. In vivo exposure of mixed microplastic particles in mice and its impacts on the murine gut microbiome and metabolome. Toxicol. Sci. 209, kfaf145 (2025). PubMed
- 46. Kuai, Y. et al. Long-term exposure to polystyrene microplastics reduces macrophages and affects the microbiota–gut–brain axis in mice. Toxicology 509, 153951 (2024). PubMed
- 47. Li, B. et al. Polyethylene microplastics affect the distribution of gut microbiota and inflammation development in mice. Chemosphere 244, 125492 (2020). PubMed
- 48. Li, H. et al. Polystyrene microplastics exposure: Disruption of intestinal barrier integrity and hepatic function in infant mice. Ecotoxicol. Environ. Saf. 288, 117357 (2024). PubMed
- 49. Li, Z., Li, Y., Cao, F., Huang, J. & Gao, X. Gut microbiota and metabolic health risks from chronic low-dose microplastic exposure with focus on Desulfovibrio spp. Ecotoxicol. Environ. Saf. 302, 118721 (2025). PubMed
- 50. Li, Y. et al. Combined exposure to microplastics and tetracycline leads to impaired skeletal development in young mice by the microbiota-gut-bone axis. Ecotoxicol. Environ. Saf. 306, 119308 (2025). PubMed
- 51. Li, M. et al. Short-term microplastic exposure: A double whammy to lung metabolism and fecal microflora in diabetic SD rats. Ecotoxicol. Environ. Saf. 297, 118229 (2025). PubMed
- 52. Li, X., Jing, K., Song, P. & Yu, J. Aged polystyrene microplastics exacerbate cadmium-induced hepatotoxicity in zebrafish through gut-liver axis metabolic dysregulation. Environ. Chem. Ecotoxicol. 7, 859–871 (2025).
- 53. Liang, X., Wang, Y., Andrikopoulos, N., Ke, P. C. & Li, Y. Dysfunctional digestive tract highlights the metabolic hallmarks of nanoplastic-exacerbated Parkinson’s pathology. npj Park.’s Dis. 11, 300 (2025). PubMed
- 54. Lu, L., Wan, Z., Luo, T., Fu, Z. & Jin, Y. Polystyrene microplastics induce gut microbiota dysbiosis and hepatic lipid metabolism disorder in mice. Sci. Total Environ. 631, 449–458 (2018). PubMed
- 55. Lu, T. et al. Chronic exposure to polyethylene terephthalate microplastics induces gut microbiota dysbiosis and disordered hepatic lipid metabolism in mice. Ecotoxicol. Environ. Saf. 298, 118330 (2025). PubMed
- 56. Marana, M. H. et al. Plastic nanoparticles cause mild inflammation, disrupt metabolic pathways, change the gut microbiota and affect reproduction in zebrafish: A full generation multi-omics study. J. Hazard. Mater. 424, 127705 (2022). PubMed
- 57. Nissen, L. et al. Single exposure of food-derived polyethylene and polystyrene microplastics profoundly affects gut microbiome in an in vitro colon model. Environ. Int. 190, 108884 (2024). PubMed
- 58. Nugrahapraja, H. et al. Effects of Microplastic on Human Gut Microbiome: Detection of Plastic-Degrading Genes in Human Gut Exposed to Microplastics—Preliminary Study. Environments 9, 140 (2022).
- 59. Qiao, J. et al. Perturbation of gut microbiota plays an important role in micro/nanoplastics-induced gut barrier dysfunction. Nanoscale 13, 8806–8816 (2021). PubMed
- 60. Rehman, A. et al. Impacts of polystyrene nanoplastics on zebrafish gut microbiota and mechanistic insights. Ecotoxicol. Environ. Saf. 299, 118332 (2025). PubMed
- 61. Shen, W. et al. Sex-Specific Effects of Polystyrene Microplastic and Lead(II) Co-Exposure on the Gut Microbiome and Fecal Metabolome in C57BL/6 Mice. Metabolites 14, 189 (2024). PubMed
- 62. Su, Q.-L. et al. The impact of microplastics polystyrene on the microscopic structure of mouse intestine, tight junction genes and gut microbiota. PLOS ONE 19, e0304686 (2024). PubMed
- 63. Sun, H., Chen, N., Yang, X., Xia, Y. & Wu, D. Effects induced by polyethylene microplastics oral exposure on colon mucin release, inflammation, gut microflora composition and metabolism in mice. Ecotoxicol. Environ. Saf. 220, 112340 (2021). PubMed
- 64. Sun, X. et al. Polyethylene terephthalate microplastics affect gut microbiota distribution and intestinal damage in mice. Ecotoxicol. Environ. Saf. 294, 118119 (2025). PubMed
- 65. Tamargo, A. et al. PET microplastics affect human gut microbiota communities during simulated gastrointestinal digestion, first evidence of plausible polymer biodegradation during human digestion. Sci. Rep. 12, 528 (2022). PubMed
- 66. Tilves, C. et al. Associations of Plastic Bottle Exposure with Infant Growth, Fecal Microbiota, and Short-Chain Fatty Acids. Microorganisms 11, 2924 (2023). PubMed
- 67. Tu, P. et al. Deciphering Gut Microbiome Responses upon Microplastic Exposure via Integrating Metagenomics and Activity-Based Metabolomics. Metabolites 13, 530 (2023). PubMed
- 68. Wang, Z. et al. Transfer toxicity of polystyrene microplastics in vivo: Multi-organ crosstalk. Environ. Int. 202, 109604 (2025). PubMed
- 69. Wang, S. et al. The influence of microplastics on hypertension-associated cardiovascular injury via the modulation of gut microbiota. Environ. Pollut. 368, 125760 (2025). PubMed
- 70. Wang, Z. et al. Polystyrene microplastics induce potential toxicity through the gut-mammary axis. npj Sci. Food 9, 139 (2025). PubMed
- 71. Wei, G. et al. Low-dose polystyrene microplastics exposure increases susceptibility to obesity-induced MASLD via disrupting intestinal barrier integrity and gut microbiota homeostasis. Ecotoxicol. Environ. Saf. 299, 118310 (2025). PubMed
- 72. Xia, Y. et al. Effects of microplastics and tetracycline induced intestinal damage, intestinal microbiota dysbiosis, and antibiotic resistome: metagenomic analysis in young mice. Environ. Int. 199, 109512 (2025). PubMed
- 73. Xie, L. et al. Intestinal flora variation reflects the short-term damage of microplastic to the intestinal tract in mice. Ecotoxicol. Environ. Saf. 246, 114194 (2022). PubMed
- 74. Xu, M. et al. Impact of Microplastic Exposure on Blood Glucose Levels and Gut Microbiota: Differential Effects under Normal or High-Fat Diet Conditions. Metabolites 14, 504 (2024). PubMed
- 75. Yang, Y.-K.-X. et al. Effects of Polyvinyl Chloride Microplastics on the Reproductive System, Intestinal Structure, and Microflora in Male and Female Mice. Vet. Sci. 11, 488 (2024). PubMed
- 76. Zhai, Z., Yang, Y., Chen, S. & Wu, Z. Long-Term Exposure to Polystyrene Microspheres and High-Fat Diet–Induced Obesity in Mice: Evaluating a Role for Microbiota Dysbiosis. Environ. Heal. Perspect. 132, 097002 (2024). PubMed
- 77. Zhang, Z. et al. Continuous oral exposure to micro- and nanoplastics induced gut microbiota dysbiosis, intestinal barrier and immune dysfunction in adult mice. Environ. Int. 182, 108353 (2023). PubMed
- 78. Zhou, Y. et al. Gut microbiota combined with metabolome dissects long-term nanoplastics exposure-induced disturbed spermatogenesis. Ecotoxicol. Environ. Saf. 267, 115626 (2023). PubMed
- 79. Agrawal, M. et al. Micro- and nano-plastics, intestinal inflammation, and inflammatory bowel disease: A review of the literature. Sci. Total Environ. 953, 176228 (2024). PubMed
- 80. Ali, N. et al. The potential impacts of micro-and-nano plastics on various organ systems in humans. eBioMedicine 99, 104901 (2024). PubMed
- 81. Bora, S. S. et al. Microplastics and human health: unveiling the gut microbiome disruption and chronic disease risks. Front. Cell. Infect. Microbiol. 14, 1492759 (2024). PubMed
- 82. Chen, X. et al. Adverse health effects of emerging contaminants on inflammatory bowel disease. Front. Public Heal. 11, 1140786 (2023). PubMed
- 83. Covello, C., Vincenzo, F. D., Cammarota, G. & Pizzoferrato, M. Micro(nano)plastics and Their Potential Impact on Human Gut Health: A Narrative Review. Curr. Issues Mol. Biol. 46, 2658–2677 (2024). PubMed
- 84. Demarquoy, J. Microplastics and microbiota: Unraveling the hidden environmental challenge. World J. Gastroenterol. 30, 2191–2194 (2024). PubMed
- 85. Duan, J., Liu, C., Bai, X., Zhao, X. & Jiang, T. Global trends and hotspots of gastrointestinal microbiome and toxicity based on bibliometrics. Front. Microbiol. 14, 1231372 (2023). PubMed
- 86. Eichinger, J., Tretola, M., Seifert, J. & Brugger, D. Review: interactions between microplastics and the gastrointestinal microbiome. Ital. J. Anim. Sci. 23, 1044–1056 (2024).
- 87. Fournier, E. et al. Microplastics in the human digestive environment: A focus on the potential and challenges facing in vitro gut model development. J. Hazard. Mater. 415, 125632 (2021). PubMed
- 88. Garrido-Romero, M. et al. Relevance of gut microbiome research in food safety assessment. Gut Microbes 16, 2410476 (2024). PubMed
- 89. Hirt, N. & Body-Malapel, M. Immunotoxicity and intestinal effects of nano- and microplastics: a review of the literature. Part. Fibre Toxicol. 17, 57 (2020). PubMed
- 90. Huang, Z., Weng, Y., Shen, Q., Zhao, Y. & Jin, Y. Microplastic: A potential threat to human and animal health by interfering with the intestinal barrier function and changing the intestinal microenvironment. Sci. Total Environ. 785, 147365 (2021). PubMed
- 91. Jiménez-Arroyo, C., Tamargo, A., Molinero, N. & Moreno-Arribas, M. V. The gut microbiota, a key to understanding the health implications of micro(nano)plastics and their biodegradation. Microb. Biotechnol. 16, 34–53 (2023). PubMed
- 92. Mauliasari, I. R. et al. Benzo(a)pyrene and Gut Microbiome Crosstalk: Health Risk Implications. Toxics 12, 938 (2024). PubMed
- 93. Mishra, S. K. et al. Microplastics as emerging carcinogens: from environmental pollutants to oncogenic drivers. Mol. Cancer 24, 248 (2025). PubMed
- 94. Osman, A. I. et al. Microplastic sources, formation, toxicity and remediation: a review. Environ. Chem. Lett. 21, 2129–2169 (2023). PubMed
- 95. Pacher-Deutsch, C. et al. The microplastic-crisis: Role of bacteria in fighting microplastic-effects in the digestive system. Environ. Pollut. 366, 125437 (2025). PubMed
- 96. Pan, I. & Umapathy, S. Probiotics an emerging therapeutic approach towards gut-brain-axis oriented chronic health issues induced by microplastics: A comprehensive review. Heliyon 10, e32004 (2024). PubMed
- 97. Romeo, M. et al. Exploring the Classic and Novel Pathogenetic Insights of Plastic Exposure in the Genesis and Progression of Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD). Livers 5, 21 (2025).
- 98. Sinha, P., Saini, V., Varshney, N., Pandey, R. K. & Jha, H. C. The infiltration of microplastics in human systems: Gastrointestinal accumulation and pathogenic impacts. Heliyon 11, e42606 (2025). PubMed
- 99. Souza-Silva, T. G. de et al. Impact of microplastics on the intestinal microbiota: A systematic review of preclinical evidence. Life Sci. 294, 120366 (2022). PubMed
- 100. Thin, Z. S., Chew, J., Ong, T. Y. Y., Ali, R. A. R. & Gew, L. T. Impact of microplastics on the human gut microbiome: a systematic review of microbial composition, diversity, and metabolic disruptions. BMC Gastroenterol. 25, 583 (2025). PubMed
- 101.↑ Zolotova, N. et al. Microplastic effects on mouse colon in normal and colitis conditions: A literature review. PeerJ 13, e18880 (2025). PubMed
- 102.↑ Li, Q., Niu, X., Cai, Y., Li, L. & Xia, Z. Exposure to submicroplastics promotes the progression of nonalcoholic fatty liver disease in ApoE-deficient mice. Toxicology 515, 154137 (2025). PubMed
- 103. Tao, J. et al. Long-term exposure to polystyrene microplastics induces hepatotoxicity by altering lipid signatures in C57BL/6J mice. Chemosphere 347, 140716 (2024). PubMed
- 104. Eskandrani, A. A. et al. Palliative effect of taurine against hepatic injury induced by polystyrene microplastics through antioxidant and metabolic pathway modulation in mice. Front. Pharmacol. 16, 1665161 (2025). PubMed
- 105. Li, Q. et al. Polystyrene microplastics induce liver fibrosis and lipid deposition in mice through three hub genes revealed by the RNA-seq. Sci. Rep. 15, 2583 (2025). PubMed
- 106. Zou, H. et al. Polystyrene Microplastics Induce Oxidative Stress in Mouse Hepatocytes in Relation to Their Size. Int. J. Mol. Sci. 24, 7382 (2023). PubMed
- 107. Zhang, H. et al. Potential toxicity of microplastics on vertebrate liver: A systematic review and meta–analysis. Ecotoxicol. Environ. Saf. 286, 117166 (2024). PubMed
- 108. Mierzejewski, K. et al. Ingested PET microplastics alter the metabolomic profile of the porcine pancreas. Sci. Rep. 15, 39227 (2025). PubMed
- 109. Mierzejewski, K. et al. PET microplastics induce lipotoxicity in the porcine pancreas. BMC Genom. 27, 139 (2026). PubMed
- 110. Mierzejewski, K. et al. Oral exposure to PET microplastics induces the pancreatic immune response and oxidative stress in immature pigs. BMC Genom. 26, 578 (2025). PubMed
- 111. Li, J. et al. Oral Ingestion of Polystyrene Microplastics Aggravates Chronic Pancreatitis Through ROS Induced NF-κb/TGF-β Signaling Pathway and Alteration of Gut Microbiota. FASEB J. 39, e71223 (2025). PubMed
- 112. Sun, J. et al. Co-exposure to PVC microplastics and cadmium induces oxidative stress and fibrosis in duck pancreas. Sci. Total Environ. 927, 172395 (2024). PubMed
- 113.↑ Lolescu, B. M. et al. Adipose tissue as target of environmental toxicants: focus on mitochondrial dysfunction and oxidative inflammation in metabolic dysfunction-associated steatotic liver disease. Mol. Cell. Biochem. 480, 2863–2879 (2025). PubMed
- 114.↑ Chartres, N. et al. Effects of Microplastic Exposure on Human Digestive, Reproductive, and Respiratory Health: A Rapid Systematic Review. Environ. Sci. Technol. 58, 22843–22864 (2024). PubMed
- 115. Wang, M. et al. The hidden threat: Unraveling the impact of microplastics on reproductive health. Sci. Total Environ. 935, 173177 (2024). PubMed
- 116. Singh, A., G, N. K., Choudhury, M., Rai, P. S. & Kabekkodu, S. P. Phthalates and epigenetics: An emerging public health concern. Curr. Res. Toxicol. 9, 100267 (2025). PubMed
- 117. Hou, J. et al. Polystyrene microplastics lead to pyroptosis and apoptosis of ovarian granulosa cells via NLRP3/Caspase-1 signaling pathway in rats. Ecotoxicol. Environ. Saf. 212, 112012 (2021). PubMed
- 118. Huang, T. et al. Maternal exposure to polystyrene nanoplastics during gestation and lactation induces hepatic and testicular toxicity in male mouse offspring. Food Chem. Toxicol. 160, 112803 (2022). PubMed
- 119. Saeed, A., Akhtar, M. F., Saleem, A., Akhtar, B. & Sharif, A. Reproductive and metabolic toxic effects of polystyrene microplastics in adult female Wistar rats: a mechanistic study. Environ. Sci. Pollut. Res. 30, 63185–63199 (2023). PubMed
- 120. Zhang, Y. et al. Reproductive toxicity of microplastics in female mice and their offspring from induction of oxidative stress. Environ. Pollut. 327, 121482 (2023). PubMed
- 121. Aghaei, Z. et al. Maternal exposure to polystyrene microplastics alters placental metabolism in mice. Metabolomics 19, 1 (2022). PubMed
- 122. Fang, Q., Wang, C. & Xiong, Y. Polystyrene microplastics induce male reproductive toxicity in mice by activating spermatogonium mitochondrial oxidative stress and apoptosis. Chem.-Biol. Interact. 396, 111043 (2024). PubMed
- 123. Zangene, S., Morovvati, H., Anbara, H., Khan, M. A. H. & Goorani, S. Polystyrene microplastics cause reproductive toxicity in male mice. Food Chem. Toxicol. 194, 115083 (2024). PubMed
- 124. Li, T. et al. Polyethylene Terephthalate Microplastic Exposure Induced Reproductive Toxicity Through Oxidative Stress and p38 Signaling Pathway Activation in Male Mice. Toxics 12, 779 (2024). PubMed
- 125. Wu, D., Zhang, M., Bao, T. T. & Lan, H. Long-term exposure to polystyrene microplastics triggers premature testicular aging. Part. Fibre Toxicol. 20, 35 (2023). PubMed
- 126. Zhao, T. et al. Prenatal and postnatal exposure to polystyrene microplastics induces testis developmental disorder and affects male fertility in mice. J. Hazard. Mater. 445, 130544 (2023). PubMed
- 127. Jin, H. et al. Chronic exposure to polystyrene microplastics induced male reproductive toxicity and decreased testosterone levels via the LH-mediated LHR/cAMP/PKA/StAR pathway. Part. Fibre Toxicol. 19, 13 (2022). PubMed
- 128. Li, S. et al. Polystyrene microplastics induce blood–testis barrier disruption regulated by the MAPK-Nrf2 signaling pathway in rats. Environ. Sci. Pollut. Res. 28, 47921–47931 (2021). PubMed
- 129. Hou, B., Wang, F., Liu, T. & Wang, Z. Reproductive toxicity of polystyrene microplastics: In vivo experimental study on testicular toxicity in mice. J. Hazard. Mater. 405, 124028 (2021). PubMed
- 130.↑ Dou, Y. et al. Lactating exposure to microplastics at the dose of infants ingested during artificial feeding induced reproductive toxicity in female mice and their offspring. Sci. Total Environ. 949, 174972 (2024). PubMed
- 131.↑ Rochman, C. M., Hoh, E., Kurobe, T. & Teh, S. J. Ingested plastic transfers hazardous chemicals to fish and induces hepatic stress. Sci. Rep. 3, 3263 (2013). PubMed
- 132. Hahladakis, J. N., Velis, C. A., Weber, R., Iacovidou, E. & Purnell, P. An overview of chemical additives present in plastics: Migration, release, fate and environmental impact during their use, disposal and recycling. J. Hazard. Mater. 344, 179–199 (2018). PubMed
- 133. Mato, Y. et al. Plastic Resin Pellets as a Transport Medium for Toxic Chemicals in the Marine Environment. Environ. Sci. Technol. 35, 318–324 (2001). PubMed
- 134. Teuten, E. L. et al. Transport and release of chemicals from plastics to the environment and to wildlife. Philos. Trans. R. Soc. B: Biol. Sci. 364, 2027–2045 (2009). PubMed
- 135. Zettler, E. R., Mincer, T. J. & Amaral-Zettler, L. A. Life in the “Plastisphere”: Microbial Communities on Plastic Marine Debris. Environ. Sci. Technol. 47, 7137–7146 (2013). PubMed
- 136. Arias-Andres, M., Klümper, U., Rojas-Jimenez, K. & Grossart, H.-P. Microplastic pollution increases gene exchange in aquatic ecosystems. Environ. Pollut. 237, 253–261 (2018). PubMed
- 137. Koelmans, A. A., Bakir, A., Burton, G. A. & Janssen, C. R. Microplastic as a Vector for Chemicals in the Aquatic Environment: Critical Review and Model-Supported Reinterpretation of Empirical Studies. Environ. Sci. Technol. 50, 3315–3326 (2016). PubMed
- 138. Liu, Y. et al. Microplastics enhance the prevalence of antibiotic resistance genes in mariculture sediments by enriching host bacteria and promoting horizontal gene transfer. Eco-Environ. Heal. 4, 100136 (2025). PubMed
- 139. Wang, Y.-F. et al. Microplastic diversity increases the abundance of antibiotic resistance genes in soil. Nat. Commun. 15, 9788 (2024). PubMed
- 140. Liu, Y. et al. Microplastics are a hotspot for antibiotic resistance genes: Progress and perspective. Sci. Total Environ. 773, 145643 (2021). PubMed
- 141. Rasool, A. et al. Interactions of microplastics with heavy metals in the aquatic environment: Mechanisms and mitigation. J. Hazard. Mater. Adv. 21, 100984 (2026); Liu, S. et al. Interactions Between Microplastics and Heavy Metals in Aquatic Environments: A Review. Front. Microbiol. 12, 652520 (2021). Link
- 142. Godoy, V., Blázquez, G., Calero, M., Quesada, L. & Martín-Lara, M. A. The potential of microplastics as carriers of metals. Environ. Pollut. 255, 113363 (2019). https://pubmed.ncbi.nlm.nih.gov/31614247/; Wu, C. et al. Effect of particle size on the colonization of biofilms and the potential of biofilm-covered microplastics as metal carriers. Sci. Total Environ. 821, 153265 (2022). PubMed
- 143. Brennecke, D., Duarte, B., Paiva, F., Caçador, I. & Canning-Clode, J. Microplastics as vector for heavy metal contamination from the marine environment. Estuar., Coast. Shelf Sci. 178, 189–195 (2016).
- 144.↑ Turner, A. & Holmes, L. A. Adsorption of trace metals by microplastic pellets in fresh water. Environ. Chem. 12, 600–610 (2015).
- 145.↑ Amereh, F. et al. Placental plastics in young women from general population correlate with reduced foetal growth in IUGR pregnancies. Environ. Pollut. 314, 120174 (2022). PubMed
- 146. Xue, J. et al. Microplastics in maternal amniotic fluid and their associations with gestational age. Sci. Total Environ. 920, 171044 (2024). PubMed
- 147. Xu, H. et al. The role of microplastics in elevated risk and tissues metabolic alterations in uterine fibroid of female patients. Environ. Pollut. 374, 126251 (2025). PubMed
- 148. Yun, X. et al. Raman-guided exploration of placental microplastic exposure: Unraveling the polymeric tapestry and assessing developmental implications. J. Hazard. Mater. 477, 135271 (2024). PubMed
- 149. Kong, F. et al. Polyethylene and Polyvinyl Chloride Nanoplastics in Human Follicular Fluid and Seminal Plasma: Impact on Fertilization and Sperm Quality. ACS Nano 19, 27159–27172 (2025). https://pubmed.ncbi.nlm.nih.gov/40689763/; Thomas, K. V., Davies, G., Okoffo, E., Wright, S. & Rauert, C. Comment on “Polyethylene and Polyvinyl Chloride Nanoplastics in Human Follicular Fluid and Seminal Plasma: Impact on Fertilization and Sperm Quality.” ACS Nano 20, 1–2 (2026). https://pubmed.ncbi.nlm.nih.gov/41530940/; Kong, F. & Tong, X. Reply to “Comment on ‘Polyethylene and Polyvinyl Chloride Nanoplastics in Human Follicular Fluid and Seminal Plasma: Impact on Fertilization and Sperm Quality”’. ACS Nano 20, 3–4 (2026). PubMed
- 150. Gan, S. et al. Therapeutic Repair of Sperm Quality Decline Caused by Polytetrafluoroethylene. Adv. Sci. 12, e05148 (2025). PubMed
- 151.↑ Zhang, C. et al. Association of mixed exposure to microplastics with sperm dysfunction: a multi-site study in China. eBioMedicine 108, 105369 (2024). PubMed
- 152.↑ Kalkman, D. N., Renkens, M. P. L. & Grundeken, M. J. Microplastics and Nanoplastics in Atheromas. N. Engl. J. Med. 390, 1726–1728 (2024). PubMed
- 153.↑ Schirinzi, G. F. et al. Cytotoxic effects of commonly used nanomaterials and microplastics on cerebral and epithelial human cells. Environ. Res. 159, 579–587 (2017). PubMed
- 154. Hwang, J. et al. Potential toxicity of polystyrene microplastic particles. Sci. Rep. 10, 7391 (2020). PubMed
- 155. Jin, W. et al. Microplastics exposure causes the senescence of human lung epithelial cells and mouse lungs by inducing ROS signaling. Environ. Int. 185, 108489 (2024). PubMed
- 156. Xu, M. et al. Internalization and toxicity: A preliminary study of effects of nanoplastic particles on human lung epithelial cell. Sci. Total Environ. 694, 133794 (2019). PubMed
- 157. Zhang, Y. et al. The potential effects of microplastic pollution on human digestive tract cells. Chemosphere 291, 132714 (2022). PubMed
- 158. Xie, S. et al. Microplastics perturb colonic epithelial homeostasis associated with intestinal overproliferation, exacerbating the severity of colitis. Environ. Res. 217, 114861 (2023). PubMed
- 159.↑ Tsochatzis, E. D. et al. Microplastics and nanoplastics: Exposure and toxicological effects require important analysis considerations. Heliyon 10, e32261 (2024). PubMed
- 160.↑ Needleman, H. L. & Gatsonis, C. A. Low-Level Lead Exposure and the IQ of Children: A Meta-analysis of Modern Studies. JAMA 263, 673–678 (1990). https://pubmed.ncbi.nlm.nih.gov/2136923/; Pocock, S. J., Smith, M. & Baghurst, P. Environmental lead and children’s intelligence: a systematic review of the epidemiological evidence. BMJ 309, 1189 (1994). PubMed
- 161. Lanphear, B. P. et al. Low-Level Environmental Lead Exposure and Children’s Intellectual Function: An International Pooled Analysis. Environ. Heal. Perspect. 113, 894–899 (2005). PubMed
- 162. Schwartz, J. Low-Level Lead Exposure and Children′s IQ: A Metaanalysis and Search for a Threshold. Environ. Res. 65, 42–55 (1994). PubMed
- 163. Colborn, T., Saal, F. S. vom & Soto, A. M. Developmental effects of endocrine-disrupting chemicals in wildlife and humans. Environ. Heal. Perspect. 101, 378–384 (1993). PubMed
- 164. Vandenberg, L. N. et al. Hormones and Endocrine-Disrupting Chemicals: Low-Dose Effects and Nonmonotonic Dose Responses. Endocr. Rev. 33, 378–455 (2012). PubMed
- 165. III, C. A. P. et al. Lung Cancer, Cardiopulmonary Mortality, and Long-term Exposure to Fine Particulate Air Pollution. JAMA 287, 1132–1141 (2002). PubMed
- 166. Dockery, D. W. et al. An Association between Air Pollution and Mortality in Six U.S. Cities. N. Engl. J. Med. 329, 1753–1759 (1993). PubMed
- 167. Grandjean, P. & Clapp, R. Perfluorinated Alkyl Substances. N. Solut.: A J. Environ. Occup. Heal. Polic. 25, 147–163 (2015). PubMed
- 168.↑ Landrigan, P. J. et al. The Lancet Commission on pollution and health. Lancet 391, 462–512 (2018). PubMed
- 169. Mensink, R. P. & Katan, M. B. Effect of Dietary trans Fatty Acids on High-Density and Low-Density Lipoprotein Cholesterol Levels in Healthy Subjects. N. Engl. J. Med. 323, 439–445 (1990). PubMed
- 170. Willett, W. C. et al. Intake of trans fatty acids and risk of coronary heart disease among women. Lancet 341, 581–585 (1993). PubMed
- 171. Mozaffarian, D., Katan, M. B., Ascherio, A., Stampfer, M. J. & Willett, W. C. Trans Fatty Acids and Cardiovascular Disease. N. Engl. J. Med. 354, 1601–1613 (2006). PubMed
- 172.Link
- 173.↑Link
- 174.Link
- 175.↑Link
- 176. Allen, S. et al. Atmospheric transport and deposition of microplastics in a remote mountain catchment. Nat. Geosci. 12, 339–344 (2019).
- 177. Amato-Lourenço, L. F. et al. Presence of airborne microplastics in human lung tissue. J. Hazard. Mater. 416, 126124 (2021). PubMed
- 178. Bhattacharya, P., Lin, S., Turner, J. P. & Ke, P. C. Physical Adsorption of Charged Plastic Nanoparticles Affects Algal Photosynthesis. J. Phys. Chem. C 114, 16556–16561 (2010).
- 179. Browne, M. A., Dissanayake, A., Galloway, T. S., Lowe, D. M. & Thompson, R. C. Ingested Microscopic Plastic Translocates to the Circulatory System of the Mussel, Mytilus edulis (L.). Environ. Sci. Technol. 42, 5026–5031 (2008). PubMed
- 180. Campanale, C., Massarelli, C., Savino, I., Locaputo, V. & Uricchio, V. F. A Detailed Review Study on Potential Effects of Microplastics and Additives of Concern on Human Health. Int. J. Environ. Res. Public Heal. 17, 1212 (2020). PubMed
- 181. Chen, Q. et al. Quantitative investigation of the mechanisms of microplastics and nanoplastics toward zebrafish larvae locomotor activity. Sci. Total Environ. 584, 1022–1031 (2017). PubMed
- 182. Shan, S., Zhang, Y., Zhao, H., Zeng, T. & Zhao, X. Polystyrene nanoplastics penetrate across the blood-brain barrier and induce activation of microglia in the brain of mice. Chemosphere 298, 134261 (2022). PubMed
- 183. Deng, Y., Zhang, Y., Lemos, B. & Ren, H. Tissue accumulation of microplastics in mice and biomarker responses suggest widespread health risks of exposure. Sci. Rep. 7, 46687 (2017). PubMed
- 184. Zhao, H.-J., Xu, J.-K., Yan, Z.-H., Ren, H.-Q. & Zhang, Y. Microplastics enhance the developmental toxicity of synthetic phenolic antioxidants by disturbing the thyroid function and metabolism in developing zebrafish. Environ. Int. 140, 105750 (2020). PubMed
- 185. Ramsperger, A. F. R. M. et al. Nano- and microplastics: a comprehensive review on their exposure routes, translocation, and fate in humans. NanoImpact 29, 100441 (2023). PubMed
- 186. Galloway, T. S., Cole, M. & Lewis, C. Interactions of microplastic debris throughout the marine ecosystem. Nat. Ecol. Evol. 1, 0116 (2017). PubMed
- 187. Huang, Y., Liu, Q., Jia, W., Yan, C. & Wang, J. Agricultural plastic mulching as a source of microplastics in the terrestrial environment. Environ. Pollut. 260, 114096 (2020). PubMed
- 188. Jin, Y. et al. Polystyrene microplastics induce microbiota dysbiosis and inflammation in the gut of adult zebrafish. Environ. Pollut. 235, 322–329 (2018). PubMed
- 189. Koelmans, A. A. et al. Microplastics in freshwaters and drinking water: Critical review and assessment of data quality. Water Res. 155, 410–422 (2019). PubMed
- 190. Koelmans, A. A. et al. Risk assessment of microplastic particles. Nat. Rev. Mater. 7, 138–152 (2022).
- 191. Kwon, J.-H. et al. Microplastics in Food: A Review on Analytical Methods and Challenges. Int. J. Environ. Res. Public Heal. 17, 6710 (2020). PubMed
- 192. Li, B. et al. Polyethylene microplastics affect the distribution of gut microbiota and inflammation development in mice. Chemosphere 244, 125492 (2020). PubMed
- 193. Li, J., Liu, H. & Chen, J. P. Microplastics in freshwater systems: A review on occurrence, environmental effects, and methods for microplastics detection. Water Res. 137, 362–374 (2018). PubMed
- 194. Lu, L., Wan, Z., Luo, T., Fu, Z. & Jin, Y. Polystyrene microplastics induce gut microbiota dysbiosis and hepatic lipid metabolism disorder in mice. Sci. Total Environ. 631, 449–458 (2018). PubMed
- 195. Llewellyn, M. J. et al. Adverse adult-onset and multigenerational effects in zebrafish (Danio rerio) developmentally exposed to polystyrene nanoplastics. Environ. Pollut. 386, 127223 (2025). PubMed
- 196. Mattsson, K. et al. Brain damage and behavioural disorders in fish induced by plastic nanoparticles delivered through the food chain. Sci. Rep. 7, 11452 (2017). PubMed
- 197. Paul-Pont, I. et al. Exposure of marine mussels Mytilus spp. to polystyrene microplastics: Toxicity and influence on fluoranthene bioaccumulation. Environ. Pollut. 216, 724–737 (2016). PubMed
- 198. Prata, J. C. Airborne microplastics: Consequences to human health? Environ. Pollut. 234, 115–126 (2018). PubMed
- 199. Qiao, R. et al. Microplastics induce intestinal inflammation, oxidative stress, and disorders of metabolome and microbiome in zebrafish. Sci. Total Environ. 662, 246–253 (2019). PubMed
- 200. Ragusa, A. et al. Deeply in Plasticenta: Presence of Microplastics in the Intracellular Compartment of Human Placentas. Int. J. Environ. Res. Public Heal. 19, 11593 (2022). PubMed
- 201. Schirinzi, G. F. et al. Cytotoxic effects of commonly used nanomaterials and microplastics on cerebral and epithelial human cells. Environ. Res. 159, 579–587 (2017). PubMed
- 202. Stock, V. et al. Uptake and effects of orally ingested polystyrene microplastic particles in vitro and in vivo. Arch. Toxicol. 93, 1817–1833 (2019). PubMed
- 203. Thompson, R. C. et al. Lost at Sea: Where Is All the Plastic? Science 304, 838–838 (2004). PubMed
- 204. Vethaak, A. D. & Legler, J. Microplastics and human health. Science 371, 672–674 (2021). PubMed
- 205. Wright, S. L. & Kelly, F. J. Plastic and Human Health: A Micro Issue? Environ. Sci. Technol. 51, 6634–6647 (2017). PubMed
- 206. Yong, C. Q. Y., Valiyaveetill, S. & Tang, B. L. Toxicity of Microplastics and Nanoplastics in Mammalian Systems. Int. J. Environ. Res. Public Heal. 17, 1509 (2020). PubMed
- 207. Zhang, Y. et al. Atmospheric microplastics: A review on current status and perspectives. Earth-Sci. Rev. 203, 103118 (2020).
- 208. Bexeitova, K. et al. Microplastics in freshwater systems: A review of classification, sources, and environmental impacts. Chem. Eng. J. Adv. 20, 100649 (2024).
- 209. Zhang, Q. et al. A Review of Microplastics in Table Salt, Drinking Water, and Air: Direct Human Exposure. Environ. Sci. Technol. 54, 3740–3751 (2020). PubMed